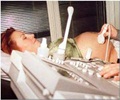
New device developed can help treat diabetic foot ulcers by sending acoustic shock waves to accelerate the healing of foot ulcers in diabetic patients.
Highlights
- New device developed helps treat diabetic foot ulcers with shock wave technology
- Dermapace System, is the device that has been approved by the US Food and Drug Administration (FDA)
- Dermapace System mechanically stimulates the wound and promotes healing
The Dermapace System has been developed by the US Sanuwave Health company. Previously, the device was approved outside of the US for advanced wound care indications.
TOP INSIGHT
The US Food and Drug Administration (FDA) approved the Dermapace System for treating chronic diabetic foot ulcers that measure more than 16 cm2.
The device functions by accelerating shock waves externally that aids in healing foot ulcers in diabetic patients. It was intended as a supplemental therapy for adult patients who are at least 22 years old and have diabetic foot ulcers, which has lasted for more than 30 days.
Dermapace System Therapy
The study is a two multicenter, randomized, double-blind trials that had about 336 patients with diabetes.
Some patients receive the usual care for ulcers like wet-to-dry dressings or debridement and also the Dermapace System therapy, while others receive the general care and a non-working shock wave therapy.
The adverse effects that were commonly observed include pain from device application, local bruising and numbness, wound infection, cellulitis, migraines, osteomyelitis, nausea, fainting, and fever.
Diabetes causes damage to the nerves and blood vessels in the feet, which makes it difficult-to-treat infections. Later, when circulation becomes poor, amputation needs to be done.
“Diabetes is the leading cause of lower limb amputations,” said Binita Ashar, MD, director of the division of surgical devices in the FDA’s Center for Devices and Radiological Health.
“The FDA is dedicated to making technologies available that can help improve the quality of life for those with chronic diseases. Additional options for successfully treating and healing ulcer wounds may help prevent lower limb amputations,” said Dr. Ashar.
The FDA has approved Dermapace System through its de novo premarket review pathway. This was designated for low-to-moderate-risk devices of a new type and cannot be matched to any other marketed devices.
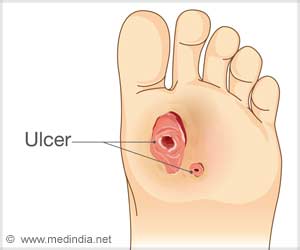
What are Foot Ulcers?
A foot ulcer caused by trauma to the foot can lead to nerve damage and blood circulation problems. Chronic foot ulcers mainly affect the elderly and when associated with poorly controlled medical problems, can lead to amputation.
Every year, it is estimated that around 4 million people would get a foot ulcer.
Diabetic ulcers have a callus, thickened and raised tissue surrounding the ridge of the ulcer. Stasis ulcers are red, shallow, with irregular edges; with a crust of necrotic tissue overlying the ulcer bed.
Diabetic foot ulcer is a major complication of Type I & 2 diabetes. In fact it is estimated that one in every six people with diabetes will have a foot ulcer during their lifetime.
Diabetic foot ulcers usually occur in the pressure areas of the feet. The bottom of the foot, esp head of the second and fifth metatarsal bones. Diabetic foot ulcers can affect one’s quality of life.
Foot problems such as ulcerations, infections, and gangrene, are the most common cause of hospitalization among diabetic patients.
Reference:
-
Foot Ulcers - (https://www.diabetes.co.uk/diabetes-complications/diabetic-foot-ulcers.html)
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email