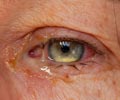
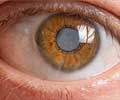
A light-sensitive lens and an ultraviolet emitting device can improve vision following cataract surgery in patients with astigmatism.
- A new lens inserted in the eye following cataract surgery can be adjusted following the surgery
- This results in improved vision in the patient, which could reduce the need for spectacles or contact lenses for vision correction following the surgery
- The lens can be used in patients with pre-existing astigmatism of ≥ 0.75 diopters without macular disease
TOP INSIGHT
A light-sensitive lens and an ultraviolet emitting device can improve vision following cataract surgery in patients with astigmatism.
Following the surgical implantation of the special lens, the patient is advised to wear special spectacles so that the lens does not alter when exposed to ultraviolet light in the environment till the end of the treatment. The treatment to change the refractive power of the lens is started 17 to 21 days following the surgery and is done in three or four sittings spread over one to two weeks. In each sitting, the patient’s eye is exposed to ultraviolet light for around 40 to 150 seconds. The adjustments are made according to the patient’s feedback, and are stopped when the patient can see well.
The device was tested in a clinical trial conducted on 600 individuals who had undergone surgery for cataract and were implanted either the special lens or conventional intraocular lens. Six months following the surgery, improvements in vision were noted on an average in patients who received the ultraviolet light-sensitive lens as compared to those who received a conventional intraocular lens.
The device should however not be used in individuals in patients with a history of herpes simplex affecting the eye or in those taking medications that increase the sensitivity to ultraviolet light; these include chloroquine, hydrochlorothiazide, the antibiotics tetracyclines and lomefloxacin, psoralens, amiodarone, phenothiazines, hypericin, the non-steroidal painkillers ketoprofen and piroxicam, and methoxsalen.
“Until now, refractive errors that are common following cataract surgery could only be corrected with glasses, contact lenses or refractive surgery. This system provides a new option for certain patients that allows the physician to make small adjustments to the implanted lens during several in-office procedures after the initial surgery to improve visual acuity without glasses,” Malvina Eydelman, M.D., director of the Division of Ophthalmic, and Ear, Nose and Throat at the FDA’s Center for Devices and Radiological Health.
About Cataract
Cataract is an age-related condition affecting the eye that results in clouding of the lens and a progressive decrease or loss of vision. The treatment involves surgical removal of the lens and replacement with an artificial intraocular lens. Laser surgery has simplified the surgery to a large extent, as a result of which patients can often be discharged on the same day of the surgery.- FDA approves first implanted lens that can be adjusted after cataract surgery to improve vision without eyeglasses in some patients - (https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm586405.htm)
 MEDINDIA
MEDINDIA



 Email
Email