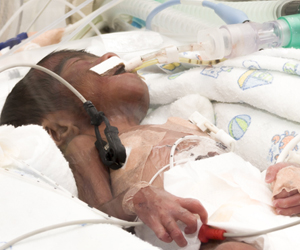
Novel device mimicking the fetal fluid filled environment within the womb could offer preterm infants the few precious weeks needed to improve their survival.
Highlights:
- Approximately 30,000 infants are born critically preterm (less than 26 weeks gestation) in the US.
- Preterm infants have a high risk of mortality and even if they survive, they are often afflicted with longterm disability due to immaturity of their lungs and other organs.
- Current device acts as a bridge between the womb and the outside environment and can support the infants up to 28 weeks after which they have a good chance of survival.
Current neonatal care practices can improve survival of infants as young as 23 weeks weeks of gestation. These infants weigh hardly a pound (about 600 grams) and have between 30-50 percent chance of living. However, many of them have longterm disabilities due to their organ immaturity.
The program brought together the combined expertise of several specialists at CHOP, including neonatologists, respiratory therapists, fetal medicine specialists, perfusionists and others. The initial thrust for the research program came over 5 years ago from CHOP research fellow Emily Partridge, MD, PhD, who has faced the challenges of caring for severely preterm infants first hand.
Said study leader Alan W. Flake, MD, a fetal surgeon and director of the Center for Fetal Research at CHOP, “ "Our system could prevent the severe morbidity suffered by extremely premature infants by potentially offering a medical technology that does not currently exist,".
Methods of the Research
- The team of scientists took over 3 years to develop and refine their prototype to the current model. They tested the device on 6 preterm lambs having a gestational age equivalent to 23-24 weeks gestation of a human fetus.
- The device simulates life in the womb as closely as possible, based on knowledge from previous neonatal research. Unlike artificial life support systems, there is no pump to do the work of the heart or a ventilator as the fetal lungs are still immature and cannot breathe in atmospheric oxygen.
- The fetal heart pumps blood via the umbilical cord into a low-resistance external oxygenator built into the device that does the work of the mother's placenta in exchanging oxygen and carbon dioxide.
- The sealed, and sterile environment inside the system is protected from variations in temperature, light, pressure and specifically from the risk of infections.
- In addition, amniotic fluid, developed in the laboratory, flows into and out of the bag maintaining a fluid filled environment within.
Results of the Trial
The lambs showed normal breathing and swallowing, and had normal growth, neurological function and organ maturation opened their eyes, grew wool, and became more active.
Scope of the Research and Future Plans
The research team plan to reduce the size of the device to match the size of human infants which are about one third the size of fetal lambs. They plan to further evaluate and refine the system.
If the prototype is developed into a successful device to support human infants, Flake hopes that in a decade from now preterm infants will continue normal development in chambers filled with amniotic fluid rather than in incubators. This will improve the quality of life for
these infants and reduce the huge health costs to the administration due to prematurity.
Flake emphasizes that they have no plans to support fetuses less than 23 weeks since the extremely small fetal size and degree of organ immaturity will mean unacceptably huge risks. However, he added, "This system is potentially far superior to what hospitals can currently do for a 23-week-old baby born at the cusp of viability. This could establish a new standard of care for this subset of extremely premature infants."
Source-Medindia