When all other treatments to fight migraine fail, a drug called erenumab may help relieve the excruciating headache.
- When all other treatments for migraine have failed, patients may find relief with a new drug called erenumab.
- Erenumab is a monoclonal antibody, which blocks pain signals by targeting a receptor for a peptide that transmits migraine pain signals.
- In a third of the patients, erenumab reduced the average number of monthly migraine headaches by more than 50%.
TOP INSIGHT
Erenumab reduced the average number of monthly migraine headaches by more than 50 percent.
Migraine and Erenumab
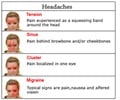
Migraine is a common neurological disorder that is often difficult to treat. It can cause severe throbbing pain or a pulsing sensation, on one or both sides of the head. A migraine may also be accompanied by nausea, vomiting, and extreme sensitivity to light and sound. It can last for a few hours to a few days and severely affects the quality of life. The drug tested in this study, Erenumab, is a monoclonal antibody that blocks pain signals by targeting a receptor for a peptide that transmits migraine pain signals called calcitonin gene-related peptide (CGRP). Erenumab binds to this receptor which CGRP would normally bind to and thereby blocks pain signals.Study overview
The study included 246 people who had episodic migraine. The participants were given injections of either 140 milligrams of erenumab or a placebo once a month for a three month period. Thirty nine percent of the study population had been treated unsuccessfully with two other medications, thirty eight percent with three medications and twenty three percent with four medications. The participants on average, experienced nine migraine headaches a month."The people we included in our study were considered more difficult to treat, meaning that up to four other preventative treatments hadn't worked for them," said study author Uwe Reuter, MD, of The Charité - University Medicine Berlin in Germany. "Our study found that erenumab reduced the average number of monthly migraine headaches by more than 50 percent for nearly a third of study participants. That reduction in migraine headache frequency can greatly improve a person's quality of life."
Study findings
- After the three month treatment period, patients treated with erenumab were three times more likely to have reduced their migraine days by 50% or more than those treated with placebo.
- Thirty percent of the people treated with erenumab had half the number of headaches compared to 14% of patients treated with placebo.
- Patients treated with erenumab had a greater average reduction in the number of days they had headaches and the number of days they needed to take drugs to stop the migraines.
- The safety and tolerability of erenumab were similar to placebo and none of the participants taking erenumab stopped treatment due to side effects.
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email