Rapid and cheap portable serological blood test (based on antigen antibody reaction) can detect liver cancer early, in the comfort of one’s home.
Highlights:
- Liver (hepatocellular) cancer is a leading cause of cancer-related deaths worldwide; in 2015 alone 788000 persons died due to liver cancer according to a World Health Organization (WHO) estimate
- Early diagnosis and treatment remains a challenge as the disease does not become symptomatic until the advanced stages
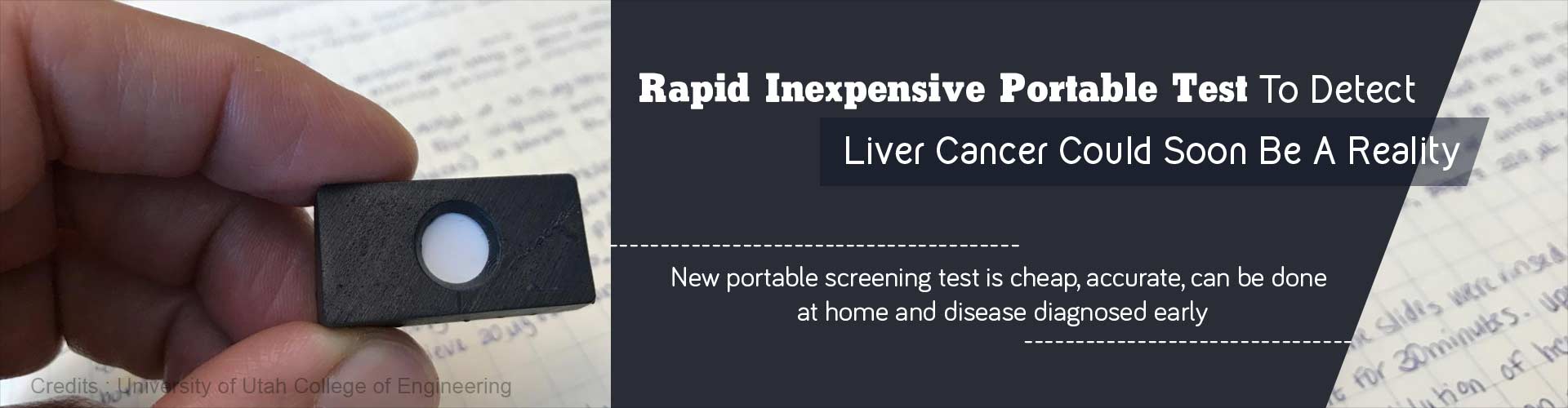
- New rapid and inexpensive accurate blood test to detect liver cancer can be done by the patient himself and overcomes costly tests and hospital visits
Rapid strides have been made in the field of cancer both in terms of diagnostic modalities available, novel treatments which have greatly improved the quality of life, as well as the long-term prognosis of patients.
TOP INSIGHT
Rapid and inexpensive test to detect liver cancer, will be a boon in low and middle income countries for mass screening and prevention, as well as follow-up of treated patients
Liver cancer is very prevalent and one of the leading causes of death in certain parts of Southeast Asia such as Mongolia. In addition, geographical considerations with lack of access to proper health care and absence of effective outreach programs also contribute to the increased mortality.
The study team believes that their simple and portable test will go a long way in early diagnosis and treatment with better outcome for such patients and their families.
Test For Early Liver Cancer Diagnosis – Alpha-fetoprotein Detection
- The test is based on the reaction of specific antigen (alpha-fetoprotein, AFP in this case) and the corresponding antibody to produce a color change or signal which is visible and can be measured.
- The test kit consists of a small domino-sized plastic cartridge having a thin paper membrane impregnated with antibody (AFP antibody here) to the specific antigen which is being tested.
- The patient’s test sample (eg: a small drop of blood, urine, saliva or even a teardrop), is dropped onto the membrane.
- Following this a drop of gold nanoparticles is added to the membrane.
- Upon adding the gold nanoparticles the trapped antigen will be tagged by the nanoparticles, resulting in a red spot on the membrane. This indicates that the person might have the disease and he needs to seek further medical advice and tests to confirm the diagnosis.
- Furthermore, a portable spectrometer (manufactured by project collaborator B&W Tek, a Delaware-based manufacturer of mobile spectrometers), can then analyze the membranes and measure the intensity of color change (proportional to the concentration of biomarkers) present. This will potentially aid in determining the severity of disease and response of patient to therapy.
"This is a smarter offshoot of that," says Porter, who also is the director of the Nano Institute of Utah.
Currently, diagnosis of liver cancer is based on blood tests and ultrasound imaging that would require travelling to a diagnostic lab or a medical facility in the city. For many persons, especially in the developing world, this could be very expensive and the results will take about two weeks.
"If we can develop a rapid test that performs at a high level of clinical accuracy," Porter says, "then we’ve got something that can have an impact on human lives."
- The team feels that the test can be modified (by changing the impregnated antibody) to diagnose several infectious diseases such as tuberculosis, dengue and malaria.
- The scientists are planning to test their technology in Mongolia in the spring of 2019, which has one of the highest death rates from liver cancer.
References:
- Third Cohort joins NCI’s Affordable Cancer Technologies Program: supporting the development of affordable technologies for cancer detection, diagnosis, and treatment in LMICs - (https://www.cancer.gov/about-nci/organization/cgh/blog/2017/acts-cohort3\
 MEDINDIA
MEDINDIA

 Email
Email