A controlled and limited market release has begun in a small number of European centers with a broad European launch scheduled for May 2015 and subsequent U.S. launch planned for the third quarter of 2015.
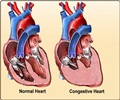
"We are excited to offer the second generation S-ICD System to physicians as a compelling treatment option for the majority of ICD-indicated patients. With the already established robust safety and efficacy clinical data, the EMBLEM S-ICD System is designed to enhance patient comfort, while still providing a less invasive treatment for patients at risk of cardiac arrest," said Kenneth Stein, M.D., Chief Medical Officer, Rhythm Management, Boston Scientific.
Source-Medindia