Midodrine, prescribed for low blood pressure, should be withdrawn, states the Food and Drug Administration as no follow-up studies have been undertaken to prove its clinical benefits.

The agency said patients who use midodrine should stop taking it and consult their doctors about other types of anti-hypotension treatment, the FDA said in a statement.
Sold under the name ProAmatine by the Irish pharmaceutical firm Shire Development Inc. and in generic versions, midodrine was approved by the FDA in 1996 based only on promising preliminary results from clinical trials.
But when granted a fast-track approval, laboratories are required to confirm the clinical benefits of the drug with post-approval tests.
To date, however, neither Shire Development nor any makers of the generic versions of midodrine have demonstrated the drug's benefits to patients suffering from orthostatic hypotension.
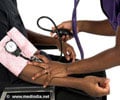
People with the condition are unable to maintain proper blood pressure in an upright position and get dizzy or faint when they stand up.
"Since the companies have not been able to provide evidence to confirm the drug's benefit, the FDA is pursuing a withdrawal of the product," he added.
If after examining data submitted by the pharmaceutical companies, the FDA continues to believe the drug should be withdrawn, it will do so, the agency said.
The generic versions of the drug are made by Apotex Corp., Impax Laboratories Inc., Mylan Pharmaceuticals, Sandoz Inc. and Upsher-Smith Laboratories, the FDA said.
The agency said about 100,000 people in the United States received prescriptions for midodrine or one of its generic versions in 2009.
In September 2009, the Government Accountability Office, an investigative arm of the US Congress, concluded in a report that the FDA should verify whether drugs authorized on a fast track basis have lived up to their initial promise.
The GAO cited ProAmatine as an example of a drug whose clinical benefits have never been established.
According to the report, the FDA has required 144 clinical studies to confirm the efficacy of drugs authorized on an fast-track basis since 1992, and to date only 64 percent of them have been carried out.
Source-AFP
 MEDINDIA
MEDINDIA



 Email
Email





