A new type of human stem cell that can develop into numerous types of specialized cells has been generated by researchers
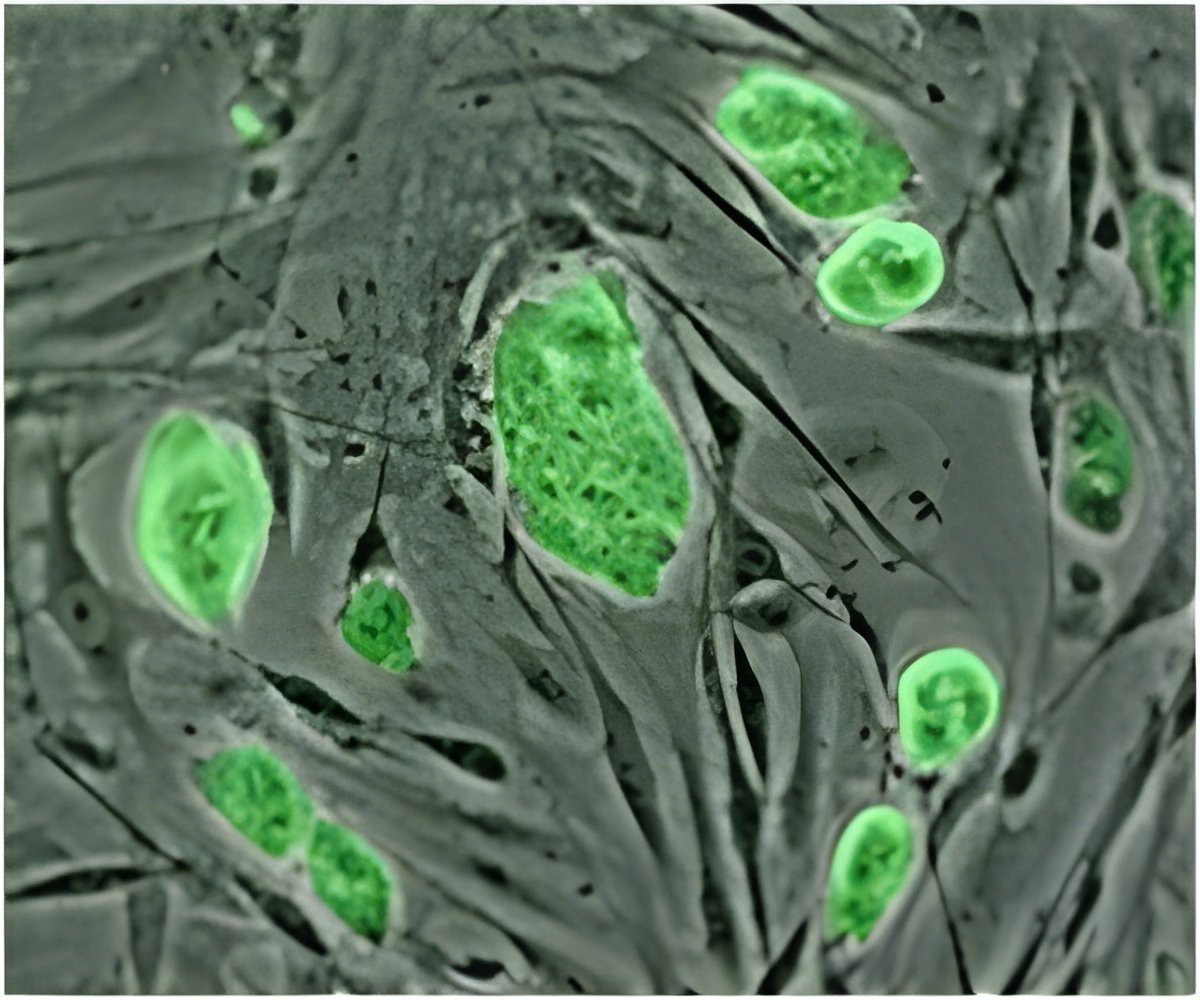
"Our cell line offers a powerful new tool for modeling how many human diseases develop," said study leader Paul J. Gadue, Ph.D., a stem cell biologist in the Center for Cellular and Molecular Therapeutics at The Children's Hospital of Philadelphia. "Additionally, pancreatic beta cells generated from EP cells display better functional ability in the laboratory than beta cells derived from other stem cell populations."
In addition to producing beta cells, the researchers also directed EP cells to develop into liver cells and intestinal cells—both of which normally develop from the endoderm tissue layer early in human development.
Gadue and colleagues are publishing their study Friday, April 6 in the journal Cell Stem Cell.
The researchers manipulated two types of human stem cells—embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs)—to become EP cells. Because both stem cell populations proliferate in great numbers and potentially generate all types of tissue, they offer enormous promise for scientists to precisely control cell development, both for the study of basic biology and for future cell-based treatments.
ESCs are derived from human embryos, typically unused embryos from fertility treatments that are donated for research purposes, while iPSCs are engineered from human somatic cells, such as skin cells or blood cells. Researchers have learned how to reprogram somatic cells to become pluripotent. Like ESCs, iPSCs are able to develop into many other types of human cells. However, when undifferentiated ESCs or iPSCs are transplanted in animal studies, they form teratomas, tumors containing many different cell types. Therefore, it has been critical that any cell type generated from ESCs or iPSCs and used for transplantation is stringently purified to exclude undifferentiated cells with tumor-forming potential.
Both in cell cultures and when transplanted into animals, the study team showed that EP cells can differentiate into multiple cell types, representing those found in the liver, pancreas and intestine. Importantly, undifferentiated EP cells did not form teratomas in the team's transplantation studies.
Gadue stressed that these promising early results are only the first steps in researching EP cells. Further work may focus on taking cells from individual patients with genetic forms of diabetes or liver disease to derive EP cell lines. The EP cell lines can then be used to model the development and progression of the patient's disease and discover new therapies for that particular disease.
Finally, although applying this science to cell therapy is years away from practical clinical use, EP cells may offer a powerful starting point for developing tissue replacement treatments, such as supplying beta cells for diabetes patients or hepatocytes (liver cells) for patients with liver disease. "While more work is needed to characterize EP cells, they may offer a potential source of safe, abundant cells for future diabetes treatments," said Gadue.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email






