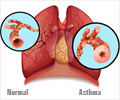
A critical cellular off switch for the inflammatory immune response that contributes to asthma attacks has been identified by researchers.
‘A critical cellular "off" switch composed of regulatory proteins that control an immune signalling mechanism in cells that contributes to asthma attacks has been identified by researchers.’





Over time, the lung is changed by secretions from the M2 cells, which cause the lung tissue to remodel itself, contributing to irreversible obstruction and poor lung function. "If you prevent these cells from becoming the M2 type, you can potentially stop the continued inflammation and long-term structural changes," said Nicola Heller, Assistant Professor at Johns Hopkins University School of Medicine in the US.
A critical cellular "off" switch for the inflammatory immune response that contributes to asthma attacks has been identified by researchers. The switch is composed of regulatory proteins that control an immune signalling mechanism in cells, the study said.
"Asthma patients are constantly firing through this pathway because those proteins are stuck in the 'on' position, without proper control by other proteins that shut down this reaction," said Heller.
The researchers believe that the findings, reported in the Journal of Biological Chemistry, also have implications for treatment of cancer and other disorders, such as obesity, in which M2 macrophage cells play a regulatory role in tumor growth and fat deposition.
Advertisement