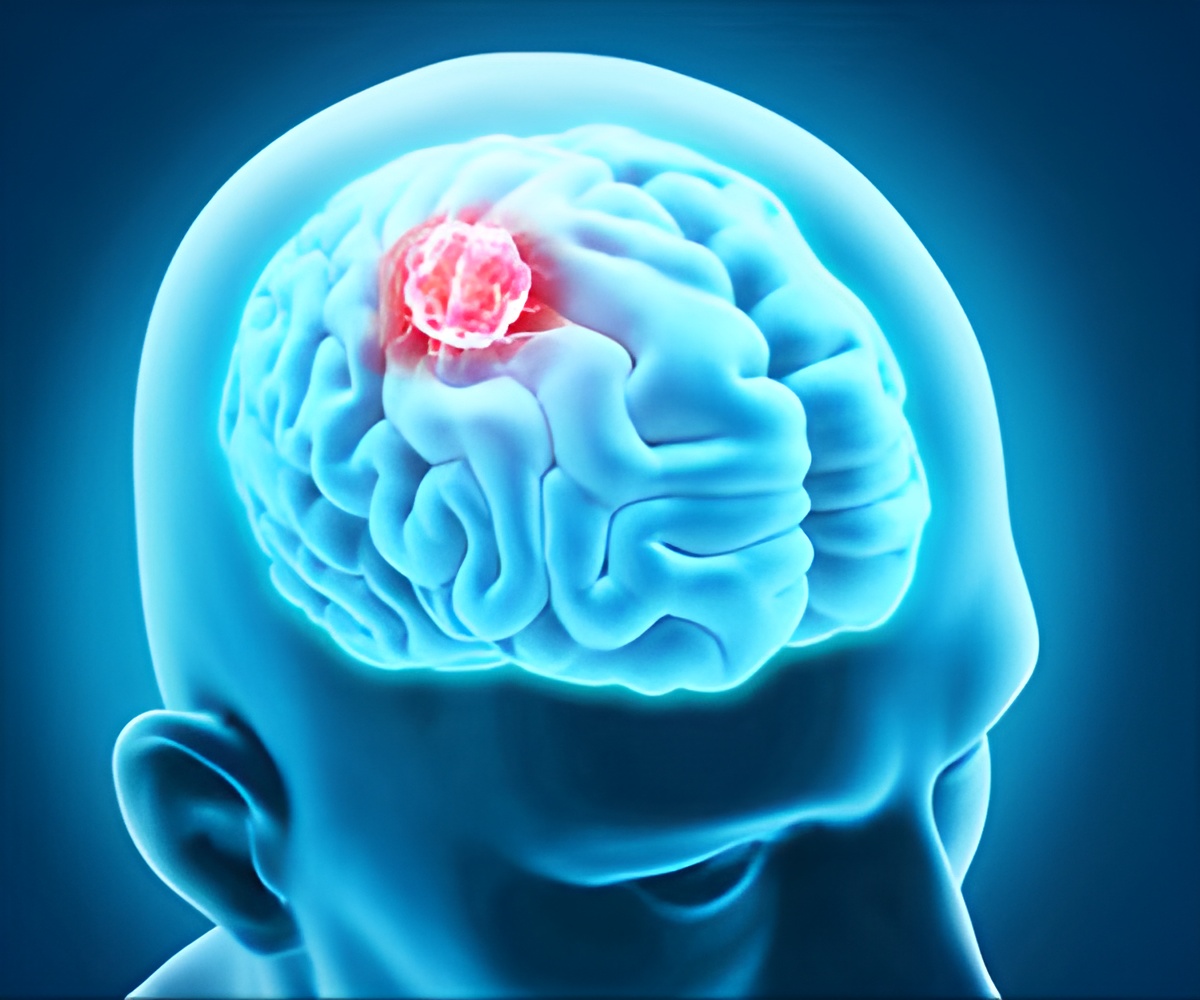
A new drug combination that stabilizes brain cancer and increases both the quantity and quality of life has been described in s new study.
TOP INSIGHT
Adding the anti-malaria drug chloroquine stopped an essential process that the cancer cells had been using to resist therapy, re-sensitizing the cancer to the targeted treatment that had previously stopped working.
"When I was 21 they found a large mass in my brain and I had it resected right away. They tested it for cancer and it came back positive," Lisa says.
"Lisa is a young adult with a very strong will to live. But it was a high-risk, aggressive glioblastoma and by the time we started this work, she had already tried everything. Miraculously, she had a response to this combination. Four weeks later, she could stand and had improved use of her arms, legs and hands," says paper first author Jean Mulcahy-Levy, investigator at the University of Colorado Cancer Center and pediatric oncologist at Children's Hospital Colorado.
The science behind the innovative, off-label use of this malaria drug, chloroquine, was in large part built in the lab of Andrew Thorburn, deputy director of the CU Cancer Center, where Mulcahy-Levy worked as a postdoctoral fellow, starting in 2009. Thorburn's lab studies a cellular process called autophagy.
From the Greek "to eat oneself," autophagy is a process of cellular recycling in which cell organelles called autophagosomes encapsulate extra or dangerous material and transport it to the cell's lysosomes for disposal. In fact, the first description of autophagy earned the 2016 Nobel Prize in Medicine or Physiology for its discoverer, Yoshinori Ohsumi.
"My initial lab studies were kind of disappointing. It didn't look like there was much effect of autophagy inhibition on pediatric brain tumors. But then we found that it wasn't no effect across the board - there were subsets of tumors in which inhibition was highly effective," Mulcahy-Levy says.
With this new understanding, Mulcahy-Levy became an essential link between Thorburn's basic science laboratory and the clinical practice of oncologist, Nicholas Foreman, MD, CU Cancer Center investigator and creator of the pediatric neuro-oncology at Children's Hospital Colorado, who had been overseeing Lisa's care.
After many surgeries, radiation treatments and chemotherapy, Lisa had started the drug vermurafenib, which was originally developed to treat BRAF+ melanoma and is now being tested in pediatric brain tumors. Lisa's experience on the drug was typical of patients with BRAF+ cancers who are treated with BRAF inhibitors such as vemurafenib - after a period of control, cancer develops additional genetic mechanisms to drive its growth and survival and is able to progress past the initial drug.
At that point, one promising strategy is to predict and/or test for new genetic dependencies and then treat any new dependency with another targeted therapy. For example, many BRAF+ cancers treated with BRAF inhibitors develop KRAS, NRAS, EGFR or PTEN changes that drive their resistance, and treatments exist targeting many of these "escape pathways". However, some cancers develop multiple resistance mechanisms and others evolve so quickly that it can be difficult to stay ahead of these changes with the correct, next targeted treatment.
"It's like that story of the boy who puts his finger in the dam," Mulcahy-Levy says. "Eventually you just can't plug all the holes."
Instead of this genetic whack-a-mole, the group chose to explore cellular mechanisms outside what can be a never-ending sequence of new mutations.
"Pre-clinical and clinical experience invariably shows that tumor cells rapidly evolve ways around inhibition of mutated kinase pathways like the BRAF pathway targeted here," the paper writes. "However, based on our results, we hypothesize that by targeting an entirely different cellular process, i.e. autophagy, upon which these same tumor cells rely, it may be feasible to overcome such resistance and thus re-establish effective tumor control."
In other words, knowing that Lisa Rosendahl's tumor was positive for BRAFV600E mutation, and that this marked the tumor as especially dependent on autophagy - and also knowing that traditional options and even clinical trials were nonexistent - the group worked with Rosendahl and her father, Greg, to add the autophagy-inhibiting drug chloroquine to her treatment.
"In September 2015, the previous targeted drugs weren't working anymore," says Greg Rosendahl. "Doctors gave Lisa less than 12 months to live. We took all our cousins up to Alaska for a final trip kind of thing. Then they came up with this new combination including chloroquine."
Vemurafenib had initially pushed Lisa's cancer past the tipping point of survival. Then the cancer had learned to use autophagy to pull itself back from the brink. Now with chloroquine nixing autophagy, vemurafenib started working again.
"My cancer got smaller, which is awesome for me," Lisa says.
"We have treated three patients with the combination and all three have had a clinical benefit. It's really exciting - sometimes you don't see that kind of response with an experimental treatment. In addition to Lisa, another patient was on the combination two-and-a-half years. She's in college, excelling, and growing into a wonderful young adult, which wouldn't have happened if we hadn't put her on this combination," Mulcahy-Levy says.
Lisa recently bought a new wheelchair so that she could spend more time at the mall. She also applied for a handicap sticker to make it easier for her to visit a nearby park with food trucks. "She wants to get out and do more. She continues to have what she feels is a good quality of life," Mulcahy-Levy says.
Research accompanying these results in patients implies that the addition of autophagy inhibition to targeted treatments may have benefits beyond glioblastoma and beyond only BRAF+ cancers. Because chloroquine has already earned FDA approval as a safe and effective (and inexpensive) treatment for malaria, the paper points out that it should be possible to "quickly test" the effectiveness of adding autophagy inhibition to a larger sample of BRAF+ glioblastoma and other brain tumor patients, and also to possibly expand this treatment to other likely mutations and disease sites.
As Mulcahy-Levy's early studies show, many cancers do not depend on autophagy. But at the same time, many do. Because a safe and simple drug already exists to inhibit autophagy, the time between discovering an autophagy-dependent cancer and the ability to add autophagy-inhibiting chloroquine to a treatment regimen against this cancer may be short.
"I really like being able to really tailor therapy to the patient," Mulcahy-Levy says. "I like saying, 'I think this is going to be really important to you,' and not necessarily using the same treatment with another patient whose cancer is driven by different genetic alterations. This is the definition of patient-centered care - designing therapy based on that individual patient's information. It's not just glioblastoma, but a certain mutation and not just the mutation but a certain pattern of previous treatments and resistance."
"It makes me feel really lucky to be a pioneer in this treatment," says Lisa Rosendahl. "I hope it helps and I hope it helps people down the road. I want it to help."
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email