A new anti-Wolbachia drug was found to have good potential to treat onchocerciasis and lymphatic filariasis which shortens the time scale of treatment from weeks to days.
TOP INSIGHT
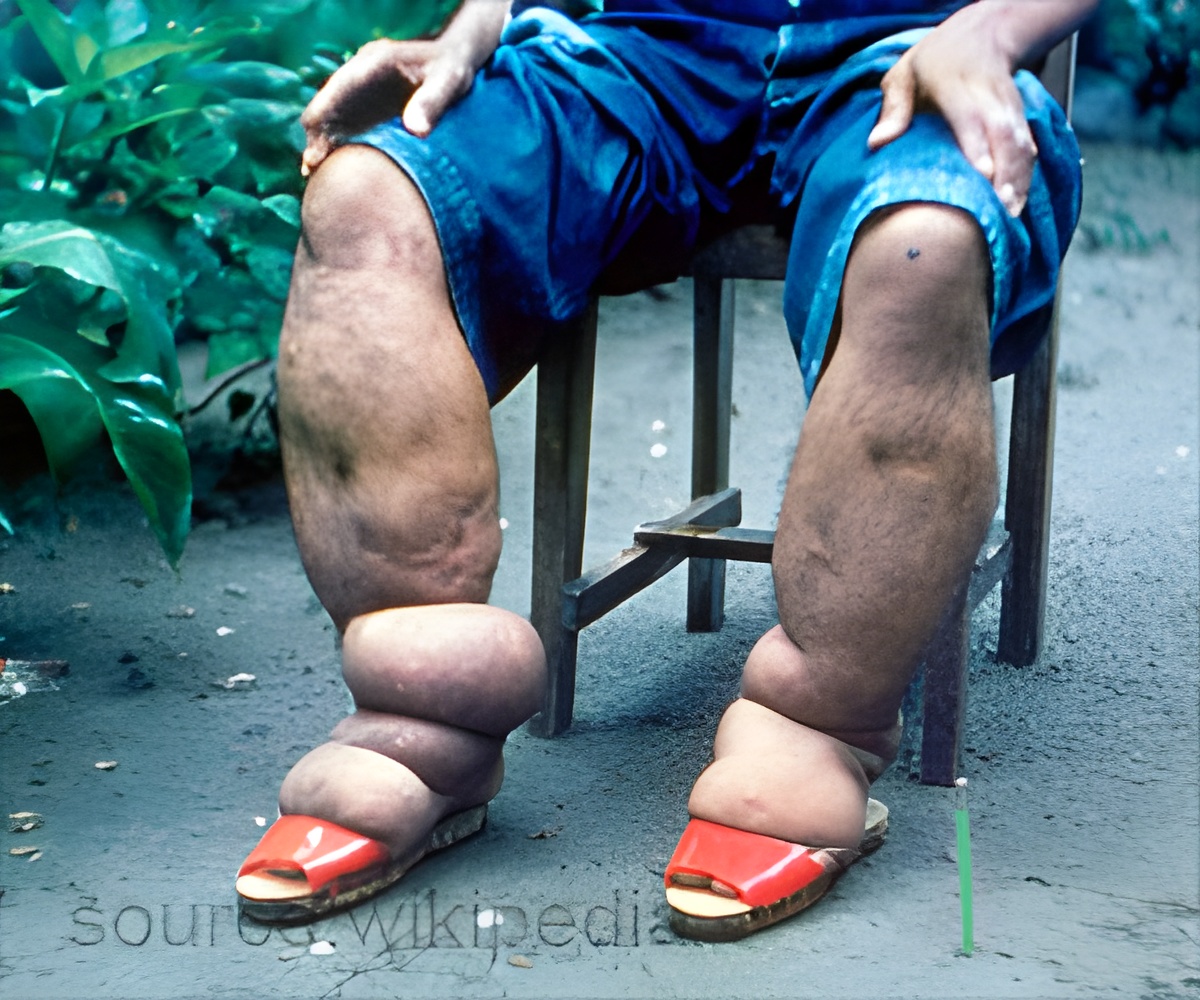
The urgent need for elimination of onchocerciasis and lymphatic filariasis could be met through a new drug compound acting against Wolbachia.
Read More..
Onchocerciasis and LF are neglected tropical diseases (NTDs) caused by filarial parasites that can lead to severe disability. The inability of current drugs to kill adult parasites has highlighted the urgent need for new curative anti-filarial drugs for these debilitating diseases.
In the search for new anti-filarial drugs, the team have focussed on targeting Wolbachia, a bacterial symbiont that is essential for parasite development and survival. Although the anti-filarial drug, doxycycline has proven effective clinically through the depletion of Wolbachia, the treatment regimens are protracted, and contradictions restrict its widespread implementation.
The starting point for the work emerged from screening activities of the A?WOL programme funded through the Bill and Melinda Gates Foundation and directed by LSTM's Professor Mark Taylor. He said: "The anti-Wolbachia strategy has proved to be a paradigm changing therapeutic approach to treatment. The repurposing of drugs already licenced and the discovery of new compounds through screening during the 10 years of A?WOL's initial work has been taken up as an alternative strategy where MDA is compromised. The further development of AWZ1066S represents another step forward in bringing relief to many millions of people."
AWZ1066S's high specificity is a significant advantage resulting in minimal impact on the gut microbiota, and the selection and emergence of resistance following the administration to patients, unlike other anti-Wolbachia based broad spectrum antibiotic treatments.
Working is close partnership with the University of Liverpool's Department of Chemistry has continued to prove a winning combination for the team. UoL's Professor Paul O'Neill is co-senior author on the paper, he said: "The novel synthetic molecule, AWZ1066S, was designed and developed at Liverpool from screening hits, and involved multi-parameter chemical optimisation with evaluation of over 400 analogues. Our approach has provided a molecule with high potency and specificity against the target pathogen along with desired oral drug properties. A key to the success of the programme was a combination of our in-house expertise in medicinal chemistry in parallel to a close working relationship with our industrial partner Eisai Ltd with additional support from AstraZeneca. This enabled development of this drug in a very short time frame"
This novel compound provides a unique opportunity to make a significant contribution to communities affected by filariasis especially if the predicted macrofilaricidal effect can be confirmed in clinical trials. These alternative strategies are needed urgently to reach the goal of onchocerciasis and LF elimination.
Source-Eurekalert
 MEDINDIA
MEDINDIA
 Email
Email