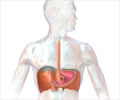
Ranitidine, the most popular acidity drug, widely used as Rantac, has found to contain a cancer-causing chemical.
TOP INSIGHT
Cancerous effect of ranitidine was noticed by the US FDA, which has issued an alert.
Ranitidine is used for multiple indications in the country and is available in different formulations, including tablets and injections. It is a prescription drug under Schedule H.
The state drug controllers have been asked to communicate the manufacturers of ranitidine to verify their products and take appropriate action for public safety.
The cancerous effect was noticed by the US FDA, which has issued an alert. Companies manufacturing the formulation in India are expected to stop production of ranitidine immediately. Following the Drugs Controller's directive, doctors would be advised by their medical associations to stop prescribing the medicine.
Source-IANS
 MEDINDIA
MEDINDIA


 Email
Email