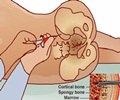
Merck announced that the late stage trial of its oral drug, Ridaforolimus successfully met its primary aim of progression-free survival among those who suffer from metastatic soft-tissue or bone sarcomas.
Named as SUCCEED, the trial was completed early this year and Merck said that the results proved that Ridaforolimus increased the progression free survival chances of the patients. The study involved patients who had displayed favorable response to chemotherapy and were given a daily 40mg dose of Ridaforolimus for five days in a week.
The researchers found that those who had taken the drug saw their progression free survival period extend to 17.7 weeks compared to 14.6 weeks among those who were placed in the placebo group.
Commenting on the study, the vice president of clinical oncology research at Merck, Eric Rubin said, “These data bring us one step closer to making ridaforolimus available to patients with metastatic sarcoma who need it and reinforces our ongoing commitment to developing innovative therapies to treat cancer.”
Source-Medindia