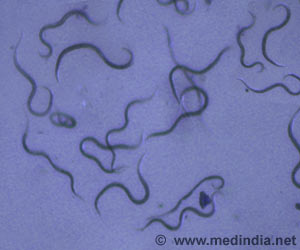
The location of the genes that control production of toxins that harm people infected by Clostridium difficile bacteria have been identified by researchers.

TOP INSIGHT
The location of the genes that control production of toxins that harm people infected by Clostridium difficile bacteria have been identified by researchers.
C. difficile resists most of the available antibiotics. "It has become important to develop a non-antibiotic therapy for this life-threatening infection," says Darkoh. "We have uncovered a pathway that we and other researchers can target to develop a non-antibiotic therapy for C. difficile infections".
C. difficile ordinarily live in the intestines - of humans, as well as animals - as one of many non-harmful bacterial species. But when a person undergoes long-term antibiotic therapy, the medicine can wipe out helpful bacterial species that keep C. difficile in check. As a result, the bacteria flourish, producing toxins and causing problems.
Some people experience only mild symptoms; for others, it's fatal. The infection causes a variety of dangerous diarrheal diseases and toxic megacolon. People in hospitals and long-term care facilities face the highest risk of infection.
CDIs are notoriously difficult to treat and infections have increased in severity and number since the beginning of the 21st century, especially among older patients. Doctors typically prescribe one of the three antibiotics, but patients become resistant to those treatments. Infections recur in as many as 25 or 30% of cases. Darkoh says C. difficile infection is a public health problem and finding a non-antibiotic method of treating CDIs is critical to public health.
Last year, in a related study published in mBio, Darkoh and his collaborators identified the accessory gene regulator (Agr) quorum signaling system as responsible for controlling synthesis of the toxins. The new study builds on that work by identifying culpable genes within the system.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email





