A new study has identified a defective process in patients with autosomal dominant polycystic kidney disease that can be targeted for preventing and treating the disease.
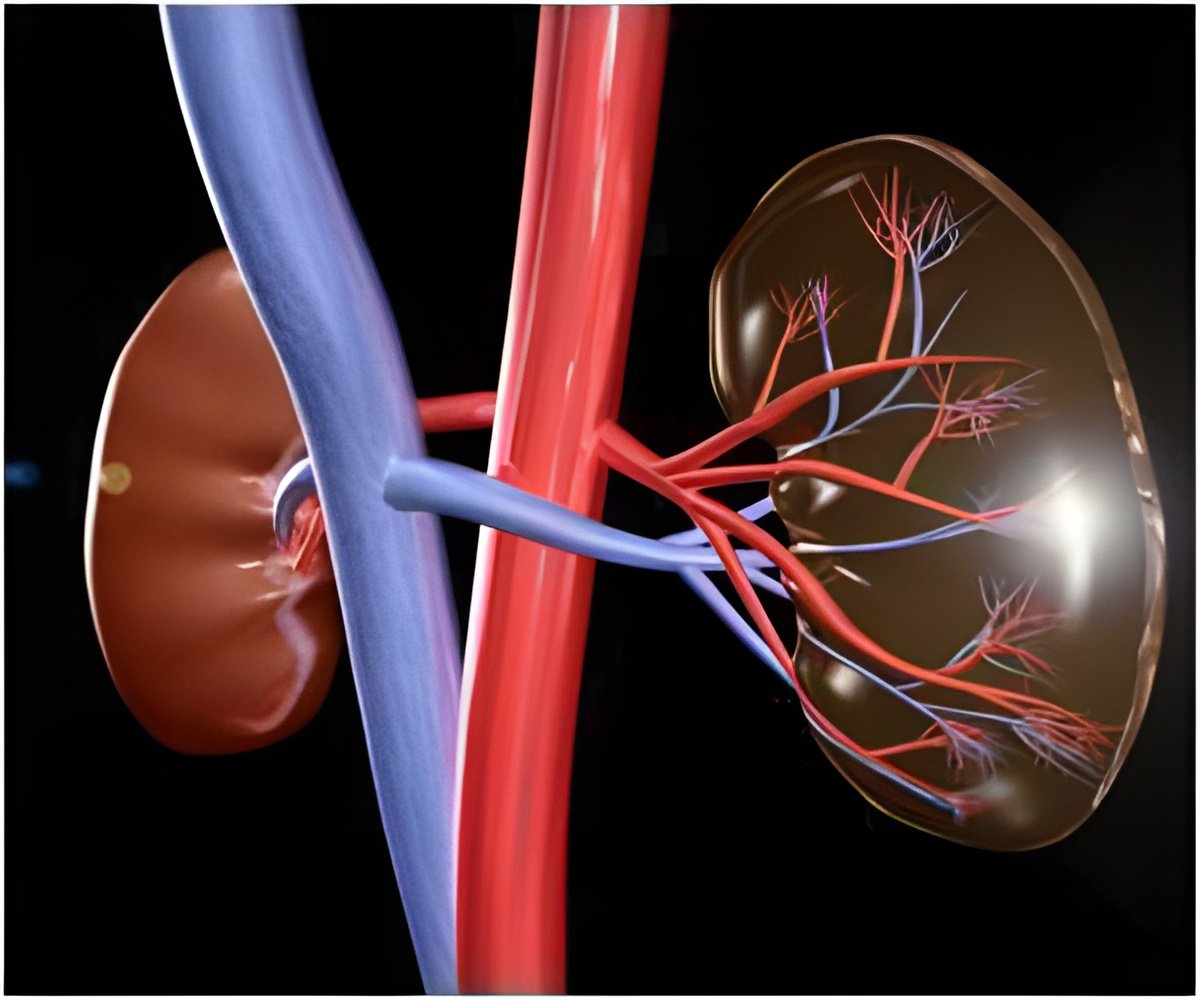
Polycystin-1, polycystin-2, and fibrocystin are all found in a cell's primary cilium, which acts as the cell's antenna and is intimately involved in human embryonic development as well as the development of certain diseases, including PKD. "What we don't know, and were hoping to better understand, is what goes wrong with these proteins in the cells of PKD patients and what kinds of therapies might help those cells," said Joseph Bonventre, MD, PhD (Brigham and Women's Hospital).
Dr. Bonventre and his colleagues Benjamin Freedman, PhD and Albert Lam, MD led a team of scientists at Brigham and Women's Hospital, the Mayo Clinic, and the Harvard Stem Cell Institute as they studied cells obtained from five PKD patients: three with ADPKD and two with ARPKD. The investigators reprogrammed patients' skin cells into induced pluripotent stem cells, which can give rise to many different cell types and tissues. When the researchers examined these cells under the microscope, they discovered that the polycystin-2 protein traveled normally to the antenna, or cilium, in cells from ARPKD patients, but it had trouble reaching the antenna in ADPKD patients. When they sequenced the DNA in these ADPKD patient cells, the investigators found mutations in the gene that encodes polycystin-1, suggesting that polycystin-1 helps shepherd polycystin-2 to the cilium.
"When we added back a healthy form of polycystin-1 to our patient cells, it traveled to the cilium and brought its partner polycystin-2 with it, suggesting a possible therapeutic approach for PKD," explained Dr. Freedman. "This was the first time induced pluripotent stem cells have been used to study human kidney disease where a defect related to disease mechanisms has been found."
The researchers noted that reprogrammed stem cells from patients with ADPKD may also be useful for testing new therapeutics before trying them out in humans.
In an accompanying editorial, Alexis Hofherr, MD and Michael Köttgen, MD (University Medical Centre, in Freiburg, Germany) stated that the study has "laid the groundwork for using induced pluripotent stem cells in PKD research. This important step forward will provide novel opportunities to model PKD pathogenesis with human cells with defined patient mutations."
Source-Eurekalert
 MEDINDIA
MEDINDIA
 Email
Email