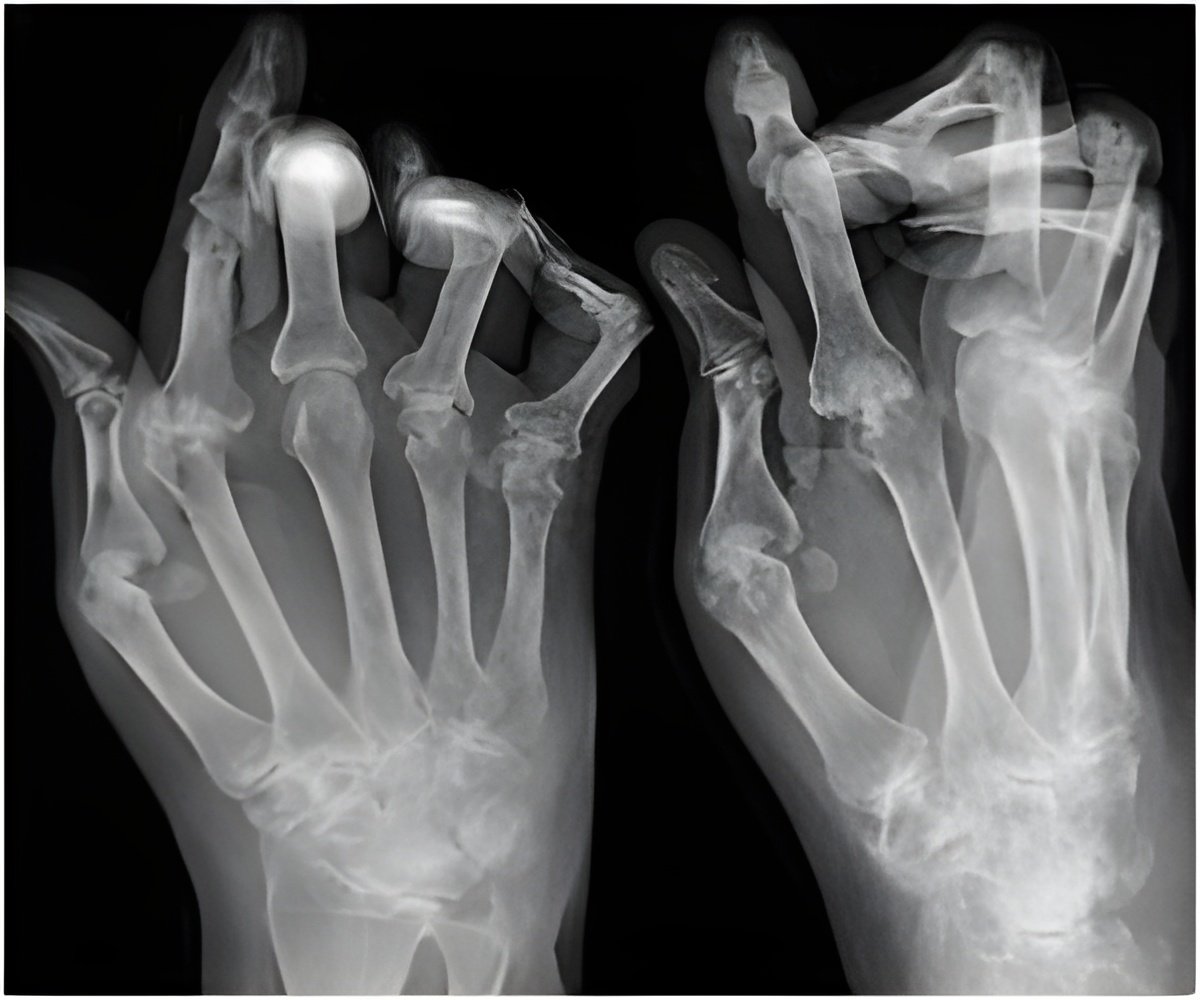
STING protein can turn down the immune response or even block its attack and help treat autoimmune diseases such as arthritis, type 1 diabetes and multiple sclerosis, say researchers.
Now they learn that the magic is in STING, which recognizes the molecule that senses the DNA then prompts release of IDO, or indoleomine 2,3-dioxyegenase.
"The fact that STING is actually part of the DNA-sensing pathway tells us something we did not know before," said Mellor, corresponding author of the study in the rapid publication section of the Journal of Immunology.
In fact, mice without STING, had no response to the DNA nanoparticle, the researchers report.
DNA nanoparticles apparently look to the body a lot like the debris that results when dying cells release DNA from their nucleus. "You could draw the analogy that DNA nanoparticles are perhaps mimicking what happens when cells die in certain ways," Mellor said. Researchers already knew there was a link between STING and immunity: the food-borne bacterium listeria releases cyclic dinucleotides to activate STING in cells it has infected. However it's the first time they realized that in this case and others, STING actually turns the immune system off. "Obviously this is one way that the bacterium may avoid being eliminated," Mellor said. He believes STING can do the same for the collagen in knee cartilage and much more.
One question is the best way to activate STING. Mellor's recently awarded $1.4 million grant from the National Institutes of Health should help answer that and more. Researchers will be comparing results from STING's activation by DNA nanoparticles to its more direct stimulation by cyclic dinucleotides. "Think of it as a surrogate for DNA," Mellor said of cyclic dinucleotide, which has a lot of appeal because it's easy to synthesize and its direct effect may reduce the side effect potential. "We need to know which is the best approach in disease," Mellor said.
A new $100,000 Arthritis Foundation grant will enable Mellor to determine if the nanoparticles also suppress collagen-induced arthritis, a widely accepted animal model for translational studies of that malady.
In the bloodstream, there are a lot of immune cells called phagocytes that ingest the submicroscopic particles that wind up in the fluid portion of the cell, called the cytoplasm, where most cellular activity happens. There, sensors detect the DNA and trigger signaling that leads to expression of IDO. In this complex interplay, STING appears essential to recognizing the molecule that recognizes the DNA.
The researchers have documented responses to their therapy in all three disease models. In the case if type 1 diabetes, for example, they have blocked the disease in a mouse genetically programmed to develop the condition.
"As long as we can treat early, they will never, ever get the disease," Mellor said. That's why his ultimate goal is vaccines. "The better we get at genetic profiling to identify individuals at risk, the more doors we open to preventing these diseases altogether, as vaccines have for so many infectious diseases. Their entire risk can be taken away to zero by early enough tweaking of the immune system."
Mellor is the Bradley-Turner and Georgia Research Alliance Eminent Scholar in Molecular Immunogenetics at MCG. He and Dr. David Munn reported in 1998 in the journal Science that the fetus expresses IDO to help avoid rejection by the mother's immune system. IDO works by degrading tryptophan, an amino acid essential to survival of T-cells, orchestrators of the immune response. Subsequent studies showed tumors use IDO for protection and the tumor-fighting potential of an IDO inhibitor is under study. On the flip side, there is evidence that increasing IDO expression can protect transplanted organs and counter autoimmune disease.
Dr. Lei Huang, an MCG research scientist, and Dr. Henrique Lemos, a Juvenile Diabetes Research Foundation-funded postdoctoral fellow, are co-authors on the published study.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email










