French drugmaker Sanofi announced that it has received the CE Mark Approval for its reusable insulin pen called JuniorSTAR.
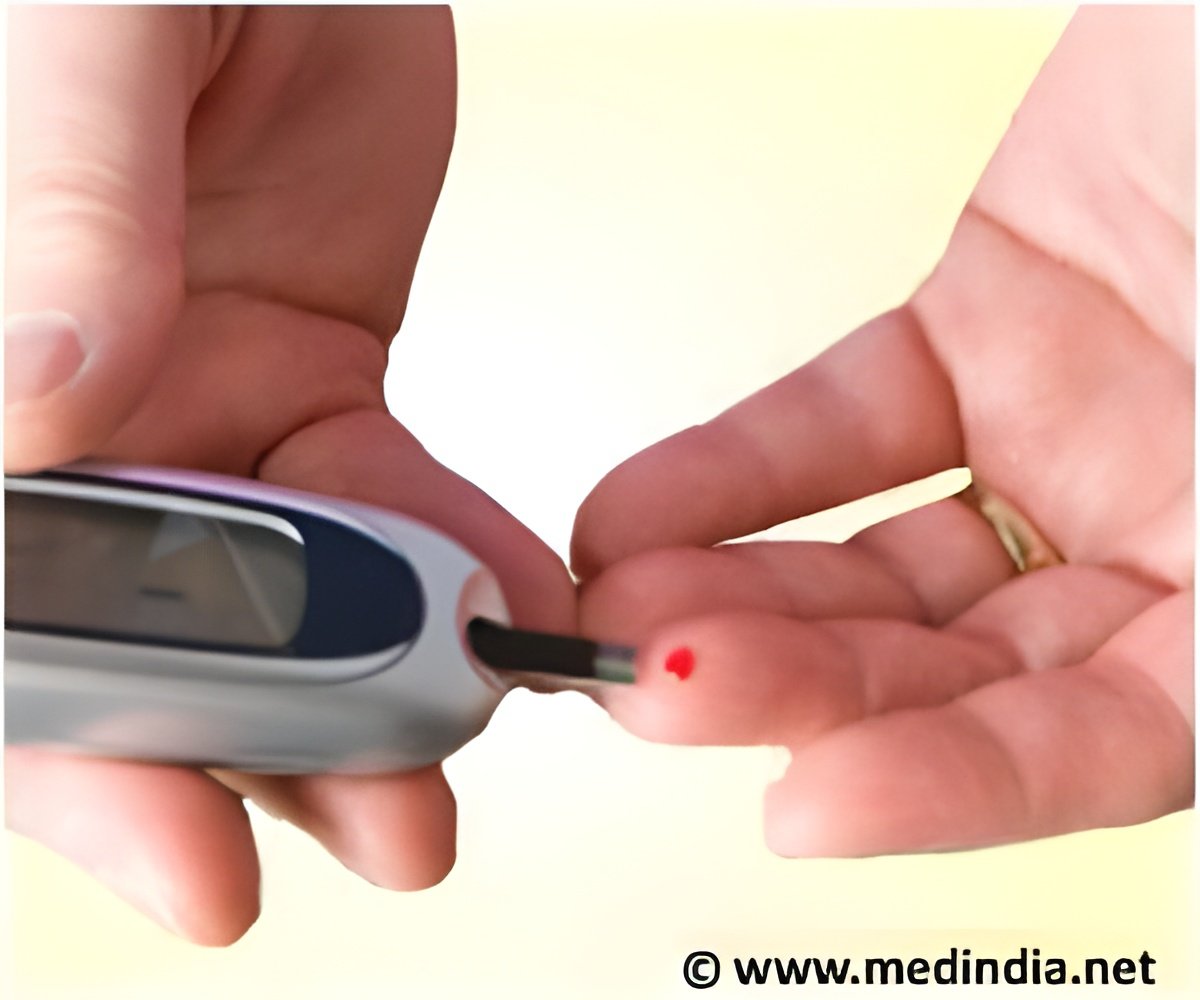
“Everyone deserves high-quality care while learning to manage their diabetes, for the best chances in life. With JuniorSTAR, we aim to meet the needs of type 1 patients worldwide, specifically the younger ones, who need an easy-to-use pen device." This is a welcome addition to our comprehensive portfolio of established insulins, which leads the way in personalized diabetes care’, the Senior Vice President, Global Diabetes, Sanofi, Pierre Chancel said.
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email









