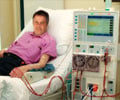
In patients with kidney failure undergoing dialysis, carbamylation protein was found to contribute to heart disease risk, say researchers.
"These modifications can make proteins inactive, or worse, can make them toxic," says Berg. "For example, when the cholesterol-carrying blood proteins LDL and HDL are carbamylated, instead of heading for tissues where they would normally be metabolized, they become attracted to atherosclerotic plaques which can lead to the development of atherosclerosis or hardening of the arteries."
Knowing that there is growing evidence that urea and protein carbamylation are important contributors to the risk of heart disease and death in patients with kidney failure, Berg and his collaborators hypothesized that measurements of carbamylated albumin - the most abundant protein in the blood --could provide an index of patients blood urea concentrations and could be used in a blood test to provide clinicians with information on how well dialysis treatments are working to remove body waste products from patients with kidney failure.
As predicted, in two independent clinical experiments, the investigators demonstrated that increased carbamylated albumin is strongly associated with an elevated risk of premature death in patients on dialysis. They additionally found that increased carbamylated albumin in dialysis patients was linked with low blood concentrations of amino acids, the building blocks of proteins.
"This suggests that amino acid deficiencies may contribute to increased protein carbamylation in dialysis patients," says Berg. "In separate experiments in both cells and in mice, we found that amino acids act as carbamylation scavengers, competitively inhibiting protein carbamylation."
The researchers were able to generate their findings through a variety of complex studies, including protein screening experiments to search for sites of carbamylation on albumin and observational studies in which they first measured albumin carbamylation in blood samples from patients with kidney disease and then followed the patients to determine whether the extent of carbamylation correlated with risk of premature death. The investigators also measured blood concentrations of amino acids in 187 patients to determine if amino acid deficiencies were linked with increased protein carbamylation. They also conducted experiments to test whether inducing amino acid deficiencies in animals led to increased protein carbamylation by urea. Finally, they conducted test tube experiments to investigate whether amino acids could directly inhibit protein carbamylation.
"Although dialysis therapy is life-saving and necessary to remove excess water and urea for patients with kidney failure, it also depletes patients'' amino acids and other essential nutrients. In this way it removes both the good and the bad, and thus simply increasing patients'' dialysis treatments will not necessarily reduce protein carbamylation," says Berg, adding that future studies will be needed to test whether amino-acid supplementation therapy reduces protein carbamylation and its associated risks, or if there are ways of modifying dialysis methods in order to remove urea without depleting amino acids.
Reference: Berg AH et al. Carbamylation of Serum Albumin as a Risk Factor for Mortality in Patients with Kidney Failure. Sci Trans Med. 2013 March 6;5(175):175ra29.
This work was funded, in part, by support from the Howard Hughes Medical Institute, the National Institutes of Health (K24 DK094872) and the University Hospital Wurzberg "Gundausstattung" grant program.
Beth Israel Deaconess Medical Center is a patient care, teaching and research affiliate of Harvard Medical School and currently ranks third in National Institutes of Health funding among independent hospitals nationwide. BIDMC is clinically affiliated with the Joslin Diabetes Center and is a research partner of the Dana-Farber/Harvard Cancer Center. BIDMC is the official hospital of the Boston Red Sox.
Source-Newswise
 MEDINDIA
MEDINDIA

 Email
Email