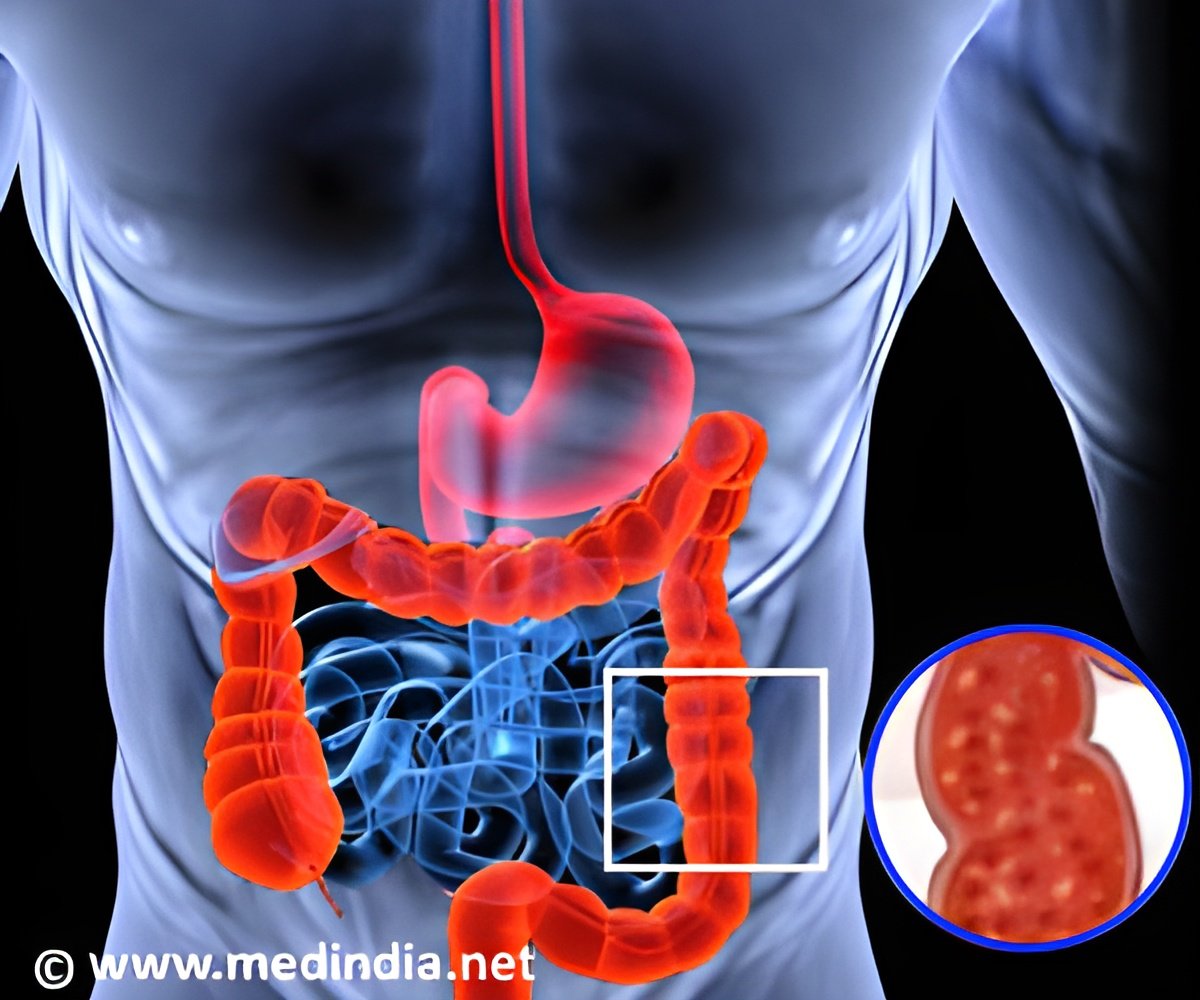
Ulcerative colitis progression can be slowed down effectively by blocking CXCL8 expression with a small molecule called repertaxin.
TOP INSIGHT
Ulcerative colitis progression can be slowed down effectively by blocking CXCL8 expression with a small molecule called repertaxin.
Read More..
Current therapy for ulcerative colitis mainly focuses on suppressing the overactive immune response with anti-inflammatory drugs. However, these therapies have limited long-term efficacy in ulcerative colitis patients.
Research has suggested that both the immune system and the ulcerative colitis microenvironment (the surrounding, less diseased cellular components) interact to drive disease development and progression.
As the immune-suppressing drugs have demonstrated limited success in treating ulcerative colitis, the researchers are studying whether targeting elements of the microenvironment—which include components of the intestinal lining (called the epithelium) and stem cell-like cells called stromal cells--may be a better or complementary treatment approach.
Lead author of the study Emina Huang, MD, staff in the Departments of Cancer Biology and Colorectal Surgery said, “Gaining a more comprehensive understanding of the complex interplay between immune and other cell types will be critical to developing new and more effective ulcerative colitis therapies and tailored, patient-specific treatment approaches.”
For the study, the researchers grew the stem cells in the lab into tiny, three-dimensional tissues that mimic actual organs (called organoids). The diseased organoids reflected histological and functional features commonly observed in ulcerative colitis patients including reduced mucus secretions, faulty barrier integrity of the intestinal lining and over expression of select proteins (including one called CXCL8).
Dr. Huang said, “We look forward to further exploring repertaxin's potential benefit in other preclinical and eventual clinical studies. We are hopeful that others will also find this model useful in identifying other potential anti-ulcerative colitis drugs.”
This approach used to develop the new ulcerative colitis model can also be used to model other complex diseases.
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email