According to results of a randomized clinical trial called RESPECT revealed that patent foramen ovale (PFO) closure may be superior to medical therapy in preventing recurrent stroke.
"As a result, the trial enrolled patients at high-risk for recurrent events and followed them for a long period of time, enabling the detection of relatively infrequent recurrent stroke," said Smalling, who is director of interventional cardiovascular medicine at the Memorial Hermann Heart and Vascular Institute. "The totality of evidence in the RESPECT trial clearly demonstrates the superiority of device closure using the Amplatzer PFO Occluder in patients with the above entry criteria compared to standard medical therapy."
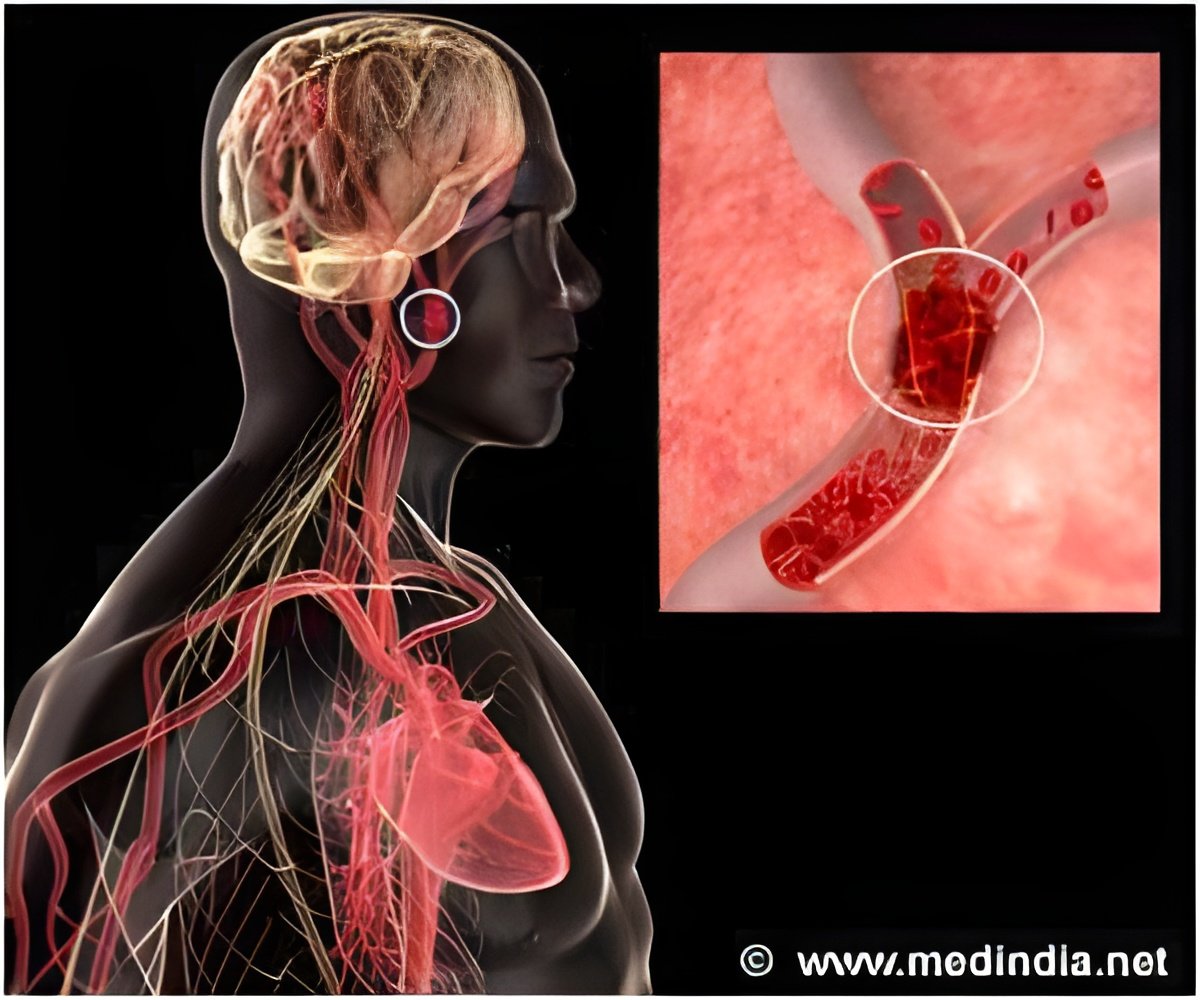
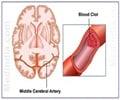
According to the National Institutes of Health, a PFO is a hole between the left and right atria (upper chambers) of the heart that fails to close naturally soon after a baby is born. In about one in four people, the hole never closes. The condition is usually not treated unless there are other heart problems or the person has a stroke caused by a blood clot. PFO has been a suspected cause of cryptogenic stroke, meaning a stroke without any identifiable cause usually occurring in people under the age of 55.
The trial enrolled 980 patients from 69 sites over eight years, yielding 2,300 patient-years of data. Medical group regimens were antiplatelet medications or warfarin. All primary endpoint events were recurrent ischemic strokes. As treated, five of the patients in the closure group had a stroke compared to 16 in the medically treated group.
"These patients with cryptogenic stroke are typically young and in the height of the productive period of their lives. Preventing a recurrent, potentially devastating, stroke by implanting a small device with very little risk is a huge potential benefit," Smalling said.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email