A bioengineered blood vessel was created by doctors at Duke University Hospital, who implanted it into the arm of a patient with end-stage kidney disease.
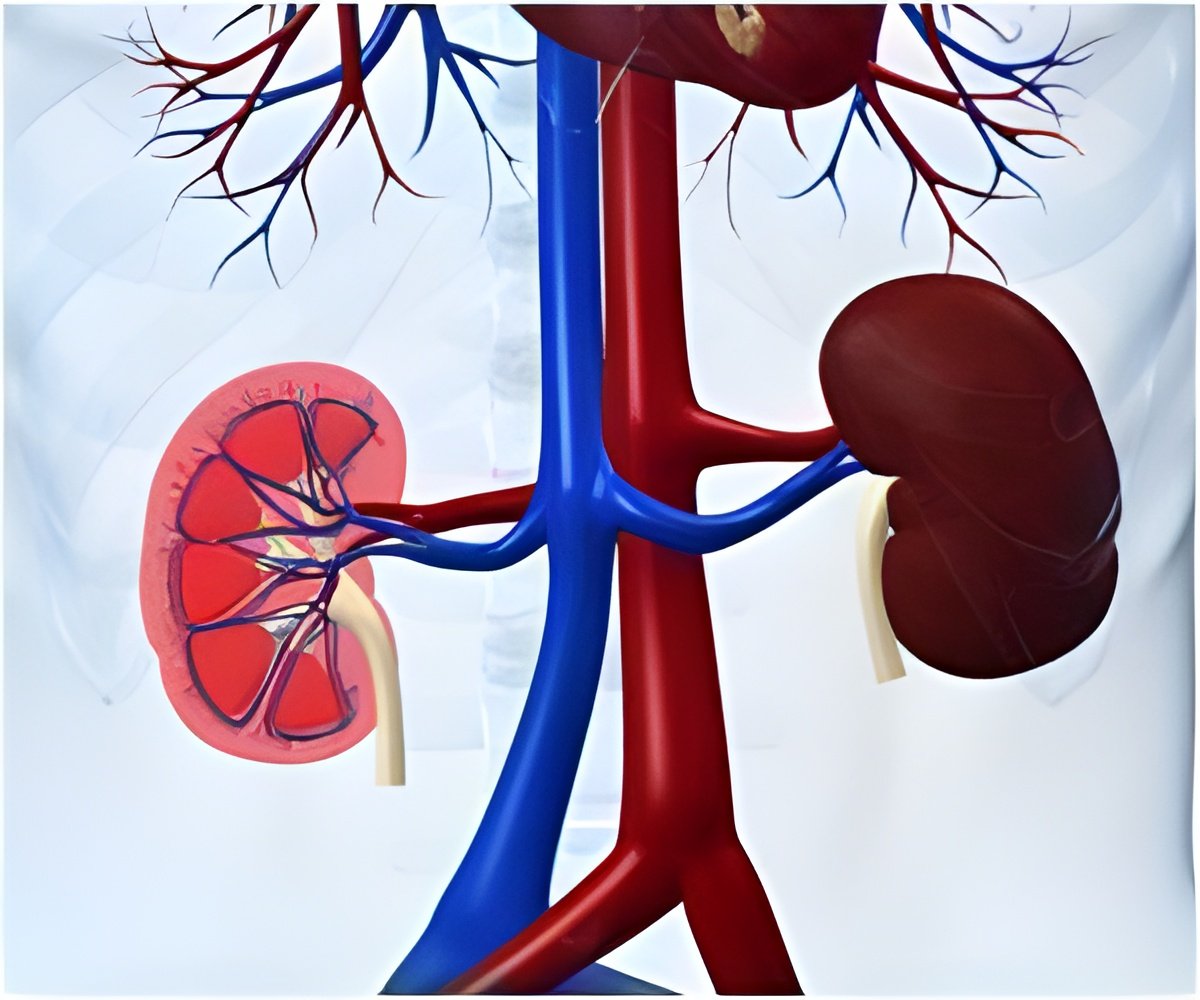
The new vein is an off-the-shelf, human cell-based product with no biological properties that would cause organ rejection.
Using technology developed at Duke and at a spin-off company it started called Humacyte, the vein is engineered by cultivating donated human cells on a tubular scaffold to form a vessel.
The vessel is then cleansed of the qualities that might trigger an immune response.
In pre-clinical tests, the veins have performed better than other synthetic and animal-based implants.
"It's exciting to see something you've worked on for so long become a reality. We talk about translational technology - developing ideas from the laboratory to clinical practice - and this only happens where there is the multi-disciplinary support and collaboration to cultivate it," Jeffrey H. Lawson, M.D., PhD, a vascular surgeon and vascular biologist at Duke Medicine who helped develop the technology and performed the implantation, said.
 MEDINDIA
MEDINDIA
 Email
Email