An ordinary protein usually involved in making other proteins is taken by the respiratory pathogen Pseudomonas aeruginosa to infest its host.
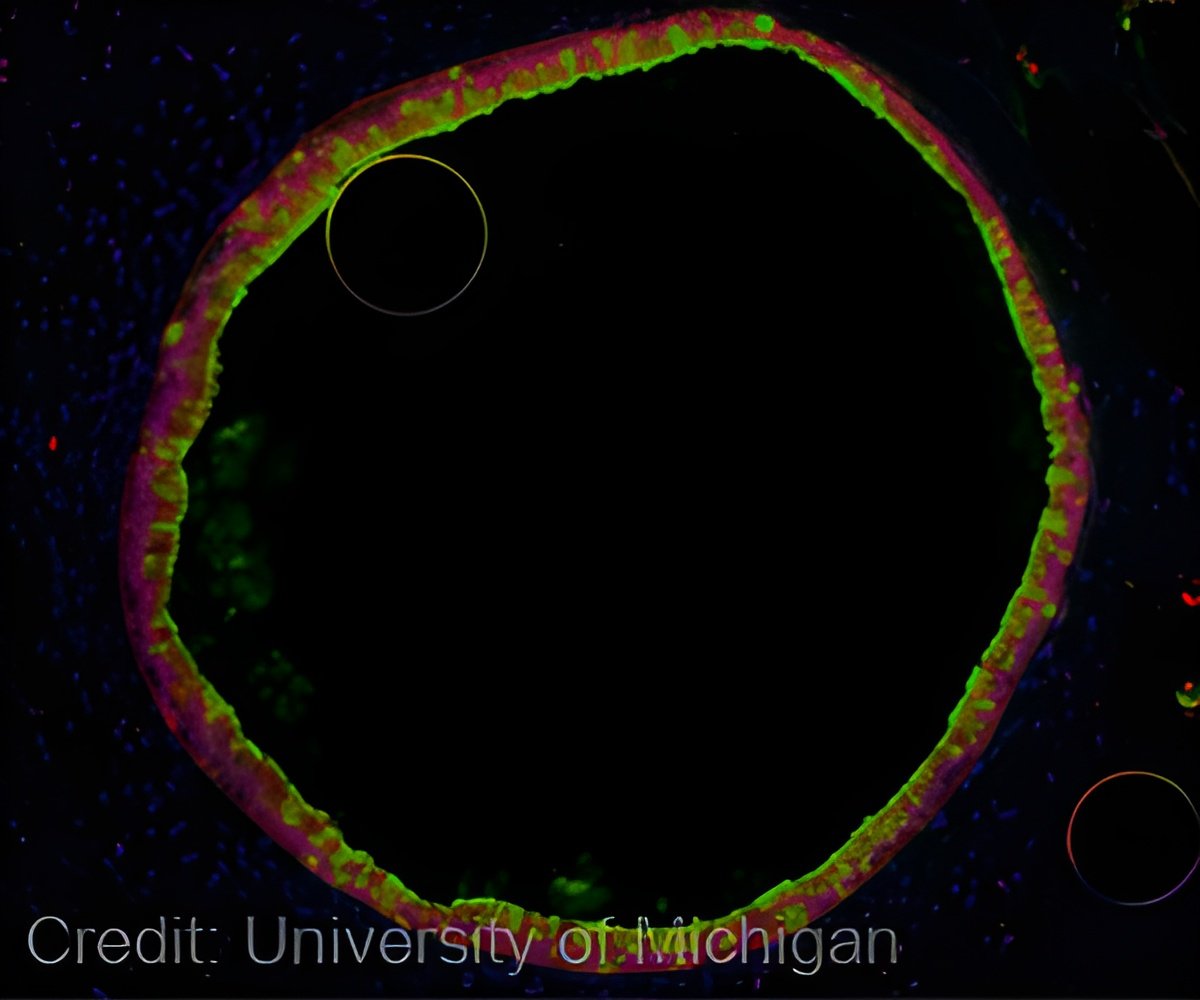
P. aeruginosa pneumonia is a big problem in the hospital setting, where it is a frequent cause of hospital-acquired pneumonia and is the leading cause of death among critically ill patients whose airways have been damaged by ventilation, trauma, or other infections. The pathogen is also a contributor to disease in the lungs of cystic fibrosis patients and forms thick biofilms that are difficult or impossible to treat with antibiotics. Goldberg and her co-author Sebastian Alberti and their colleagues study the molecular events that enable the bacterium to infect human cells in the hopes of finding ways to prevent P. aeruginosa pneumonia.
In their earlier work, Goldberg and Alberti found that P. aeruginosa takes the protein EF-Tu, which was generally thought to exist only inside the cell, and decorates the exterior of the cell with it, but in a modified form. This modified EF-Tu is recognized by antibodies to the common bacterial epitope phosphorylcholine (ChoP), indicating that the EF-Tu is modified somehow to mimic ChoP, allowing P. aeruginosa to enjoy the benefits of ChoP. By interacting with receptors on human cells, ChoP carries out a crucial step for setting up an infection for a number of different types of respiratory pathogens.
But how is EF-Tu modified, they wondered? And does it help P. aeruginosa establish an infection? This study answers those questions.
Using a host of techniques, including mass spectrometry, site directed mutagenesis of key residues in the protein, and genetic loss of function/gain of function studies, they found that P. aeruginosa only makes small changes to EF-Tu to get it to mimic this powerful ligand. P. aeruginosa transfers three methyl groups to a lysine on EF-Tu, giving it a structure similar to ChoP and allowing it to fit in the PAFR receptor in the way ChoP does.
But the modified EF-Tu not only looks like ChoP, in many ways it works like ChoP: testing in cultures of human airway cells shows that the modification of EF-Tu enables the bacterium to adhere to human cells.
As an environmental pathogen, P. aeruginosa lives in soil, water, and other environments outside the body, a fact that may offer a clue why it uses this re-purposed protein as a virulence factor. Proteins that can be put to work in both worlds - in the environment and the in the human host - would be useful to P. aeruginosa in much the way a spork can allow you to enjoy both the coleslaw and the pudding in your take out dinner.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email







