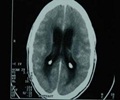
Novartis has won approval by the EU's medicine agency for its Bexsero vaccine against meningitis B infections, say sources.

"MenB disease is easily misdiagnosed, can kill within 24 hours and may cause serious, life-long disabilities," Novartis said.
The highest rates of the disease occur the first year of life, peaking by 7 months of age, the statement said.
A final decision on whether the vaccine can be launched throughout the European Union and European Economic Area must be announced within three months by the European Commission, Novartis said, indicating that EU chiefs usually followed the recommendations of the Committee for Medicinal Products for Human Use (CHMP).
Approximately 1.2 million cases of invasive meningococcal disease are recorded worldwide, and 7,000 occur in Europe, according to the European Medicines Agency (EMA) which approved the vaccine.
Of the 13 meningococcal types, EMA said, in Europe group B is the most prevalent, with 3,406-4,819 cases reported annually between 2003 and 2007.
 MEDINDIA
MEDINDIA


 Email
Email






