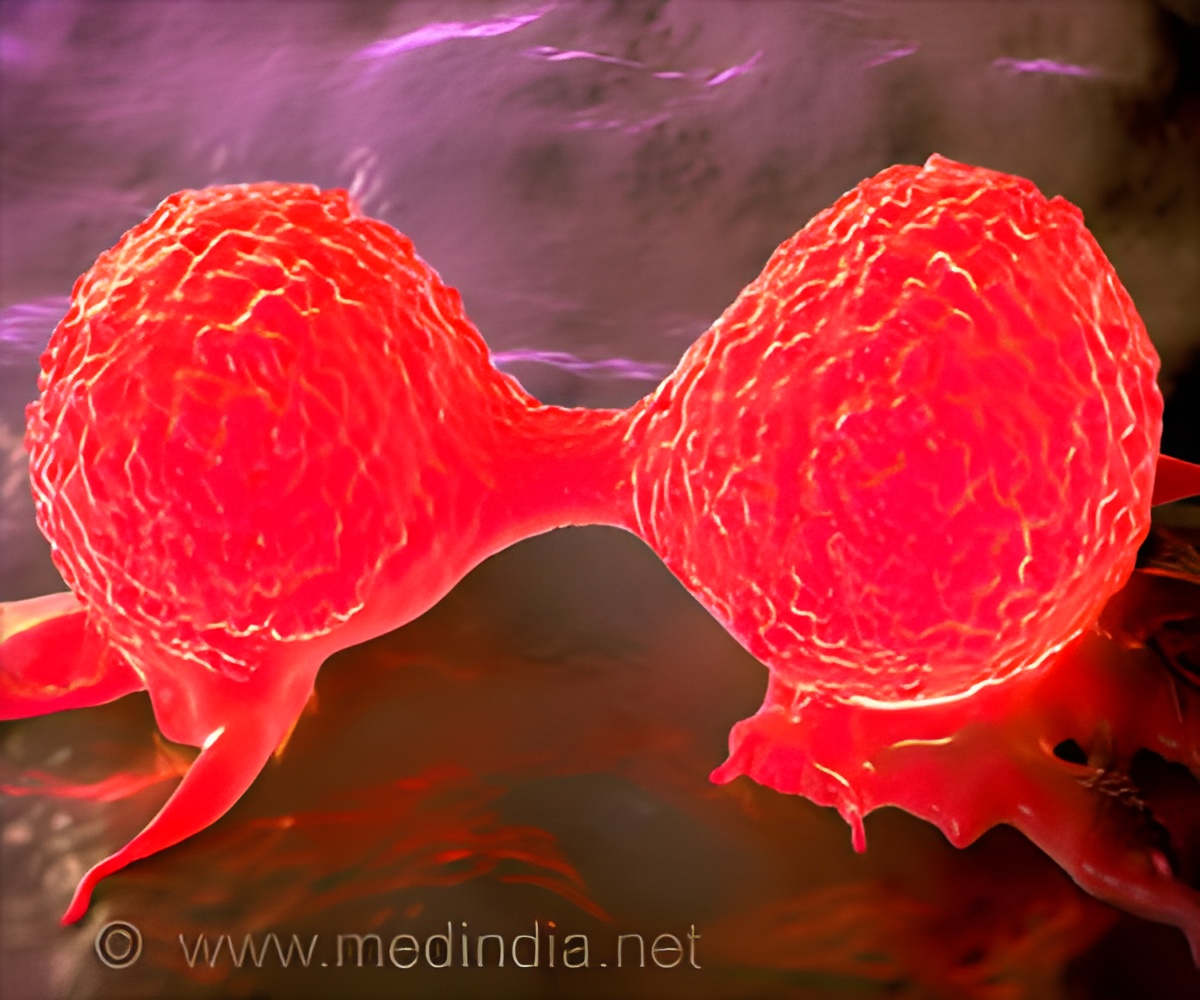
A new tool to help physicians and patients with long bone metastasis make decisions about the most appropriate treatment.
TOP INSIGHT
The new tool helps to classify patients with LBM into categories based on their survival which enables doctors to provide the right kind of treatment, avoiding overtreatment and undertreatment.
Physicians and surgeons are often called upon to estimate survival for patients with advanced cancer to help them maximize the remaining quality of life. Accurate survival estimates are important to avoid overtreatment (putting the patient through treatments that would ultimately not provide much benefit) and undertreatment (not offering treatments that would be beneficial). But data on which to base survival estimates are often sparse, especially for patients with LBMs.
To assess their "easy-to-use prognostic model," Dr. Willeumier and colleagues analyzed 1,520 patients treated for symptomatic LBMs at six Dutch hospitals between 2000 and 2013. The patients' average age was 65; the most common initial (primary) cancer sites were the breast and lung. The main symptoms requiring treatment were painful bone lesions and impending or actual pathologic fractures.
The authors previously identified three independent predictors of survival in patients with spinal metastases and applied them to these patients with LBM:
• Clinical profile of the primary tumor. Among the patients with LBM, the profile was "favorable" (longer survival) in those with primary breast cancer but "unfavorable" (shorter survival) in those with primary lung cancer.
• Presence of organ/brain metastases, in addition to LBMs.
The new model can help physicians and patients with LBMs make decisions about the most appropriate treatment, the authors believe. For example, in patients with longer expected survival, more extensive surgery might avoid failure of the implant over time and preserve function. In contrast, for those expected to live only a few months, palliative therapy might be a more appropriate choice.
The authors have developed an online and mobile app that further facilitates use of the model. The English-language version of the "OPTIModel" app can be accessed at http://optimal-study.nl/nl_NL/tooleng/.
"The simplicity and clarity of the model facilitate and encourage its use in the routine care of patients with LBM, to provide the most appropriate treatment for each individual patient," Dr. Willeumier and coauthors add. They believe their survival estimation tool might become even more useful in the future, with continued advances in tumor classification and individualized cancer therapies.
Source-Eurekalert
 MEDINDIA
MEDINDIA
 Email
Email






