Lutetium-177 (Lu-177)-labeled PSMA-617 will provide an additional therapy option for end-stage metastasized, heavily pretreated prostate cancer patients.

TOP INSIGHT
Lutetium-177 (Lu-177)-labeled PSMA-617 is a promising new therapeutic agent for radioligand therapy (RLT) of patients with metastatic castration-resistant prostate cancer.
The study is published in the January 2017 issue of the Journal of Nuclear Medicine and is the featured article. "Previous studies with small number of patients have indicated the high potential of this new therapeutic option," explains Kambiz Rahbar, University Hospital Muenster. "This study of a large number of patients at multiple healthcare facilities, however, confirms the efficacy and safety of Lu-177-PSMA-617 radioligand therapy."
At 12 therapy centers across Germany, 145 patients (median age 73 years, range 43-88) with mCRPC were treated with Lu-177-PSMA-617 between February 2014 and July 2015 with one to four therapy cycles. A total of 248 therapy cycles were performed in 145 patients.
Efficacy was defined by a prostate-specific antigen (PSA) decline of 50% or more from baseline to at least two weeks after start of RLT. Overall, 45% of patients had a positive response following all therapy cycles, while 40% responded after a single cycle.
Some adverse side effects were noted but manageable. Hematotoxicity occurred in 18 patients: 10% experienced anemia; 4%, thrombocytopenia (low platelet count); and 3%, leukopenia (low white blood cell count). Dry mouth was experienced by 8%.
"This therapeutic will provide an additional therapy option for end-stage metastasized, heavily pretreated prostate cancer patients," said Rahbar. "While already demonstrating remarkably high response rates and low toxicity, in the future it may be available at earlier stages of disease with even higher response rates and lower toxicity."
 MEDINDIA
MEDINDIA
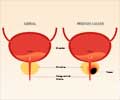
![Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis](https://images.medindia.net/patientinfo/120_100/prostate-specific-antigen.jpg)
 Email
Email