New research shows how aging can impact organs differently throughout the body.
TOP INSIGHT
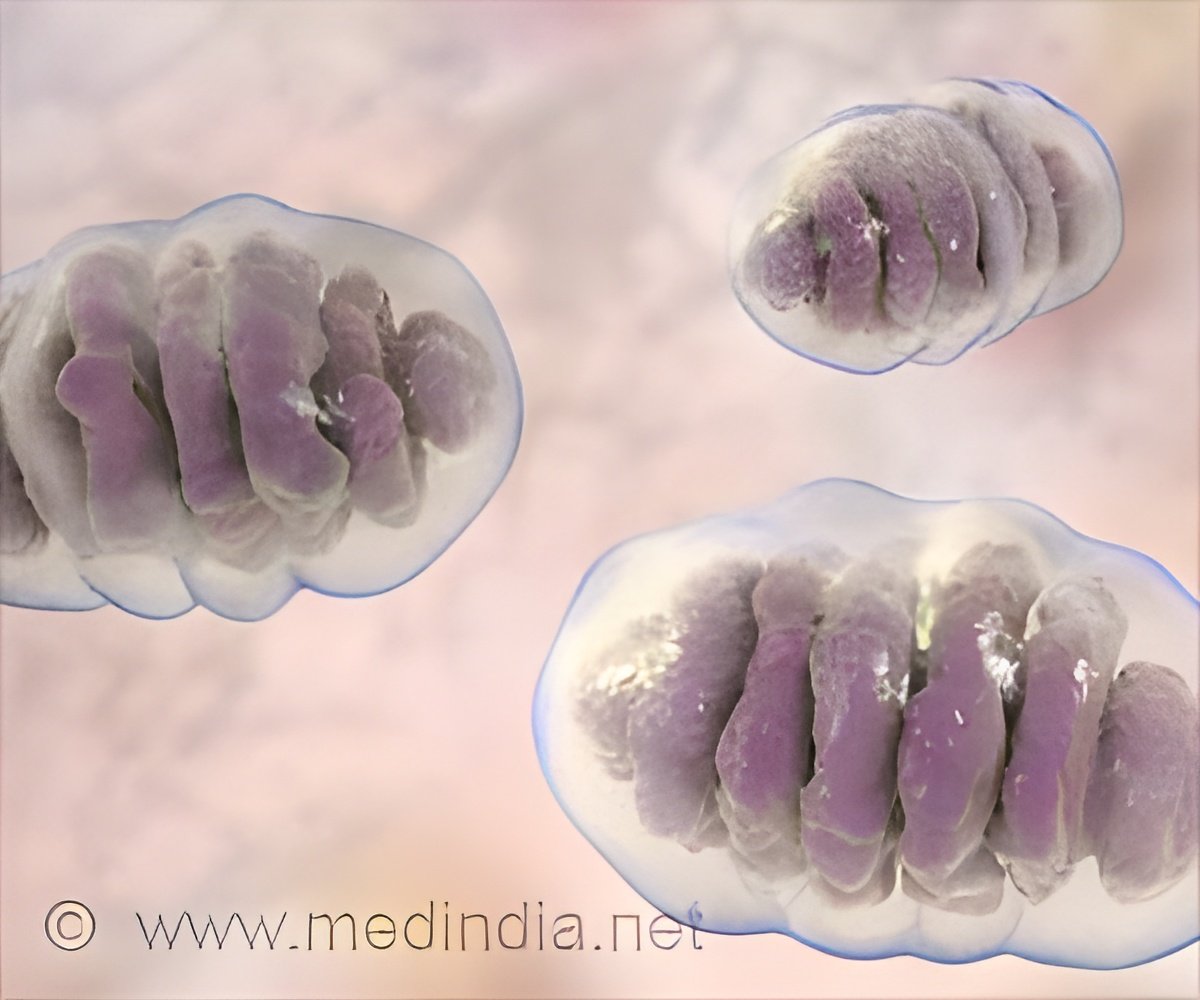
Defective energy production in old neurons might help explain why human brain is prone to age-related diseases.
Previously, the Gage lab developed a method to directly convert skin cells into neurons (called induced neurons, or iNs). Most methods to create neurons from patient cells rely on an intermediary stem cell step (creating what are called induced pluripotent stem cells), which resets cellular markers of aging. But the Gage lab's iNs retained signs of aging, including changes to gene activity and the cells' nuclei, the team reported in 2015.
In the new work, the researchers asked whether mitochondria in the cells also retained hallmarks of aging during the iN conversion process. Using skin cells collected from humans ranging in age from 0 to 89 years old, the team created iNs from each donor and then used a variety of methods to study the mitochondria of each set of cells.
Mitochondria in the skin cells isolated from each person showed few age-related changes. However, once the cells were directly converted to neurons, mitochondria from older donors were significantly different. Mitochondrial genes related to energy generation were turned off and the mitochondria were less dense, more fragmented and generated less energy.
"Pretty much every area we looked at--functional, genetic, and morphological--had defects," says Jerome Mertens, a Salk staff scientist and co-corresponding author of the new paper.
"There is no other in vitro human neuronal model to study aging," says Yongsung Kim, a research associate and first author of the paper. "So the big takeaway from our paper is that we developed a tool that enables us to study neurological aging and age-related diseases."
 MEDINDIA
MEDINDIA




 Email
Email






