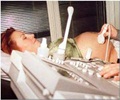
It analyzes high-resolution images of the retina in just a few hours, and uses this information to automate detection of diabetic retinopathy.

EyeArt is designed to help healthcare professionals identify patients who need immediate attention in a more effective and efficient manner.
Diabetic Retinopathy (DR), a common microvascular complication of diabetes, is the leading cause of blindness in the adult working-age population. Early detection and treatment can reduce the number of people with vision loss caused by DR by up to 90%.
Automation of diabetic retinopathy screening is the only realistic method to screen a large proportion of those with diabetes. This screening will also significantly reduce healthcare costs and improve efficiency for ophthalmologists and other eye care specialists.
Extensive clinical validation studies for EyeArt have shown that the product can achieve better sensitivity than human graders, while cutting the screening workload in half. EyeArt’s cloud-based programming interface is expected to be made widely available in Europe and the US within a month.
"Receiving the CE Marking for our flagship screening product, EyeArt, is a significant milestone for Eyenuk. Our exhaustive research and patented image analysis technology behind EyeArt has culminated in a DR screening product that is more sensitive than human graders, while still reducing workloads by more than 50%," said Dr. Kaushal Solanki, CEO of Eyenuk.
Source-Medindia
 MEDINDIA
MEDINDIA

 Email
Email