Breakdown of the neutrophil protein can cause autoimmune diseases of the blood vessels, reveals study.
TOP INSIGHT
Destroying the neutrophil protein can cause severe autoimmune disease of the blood vessels
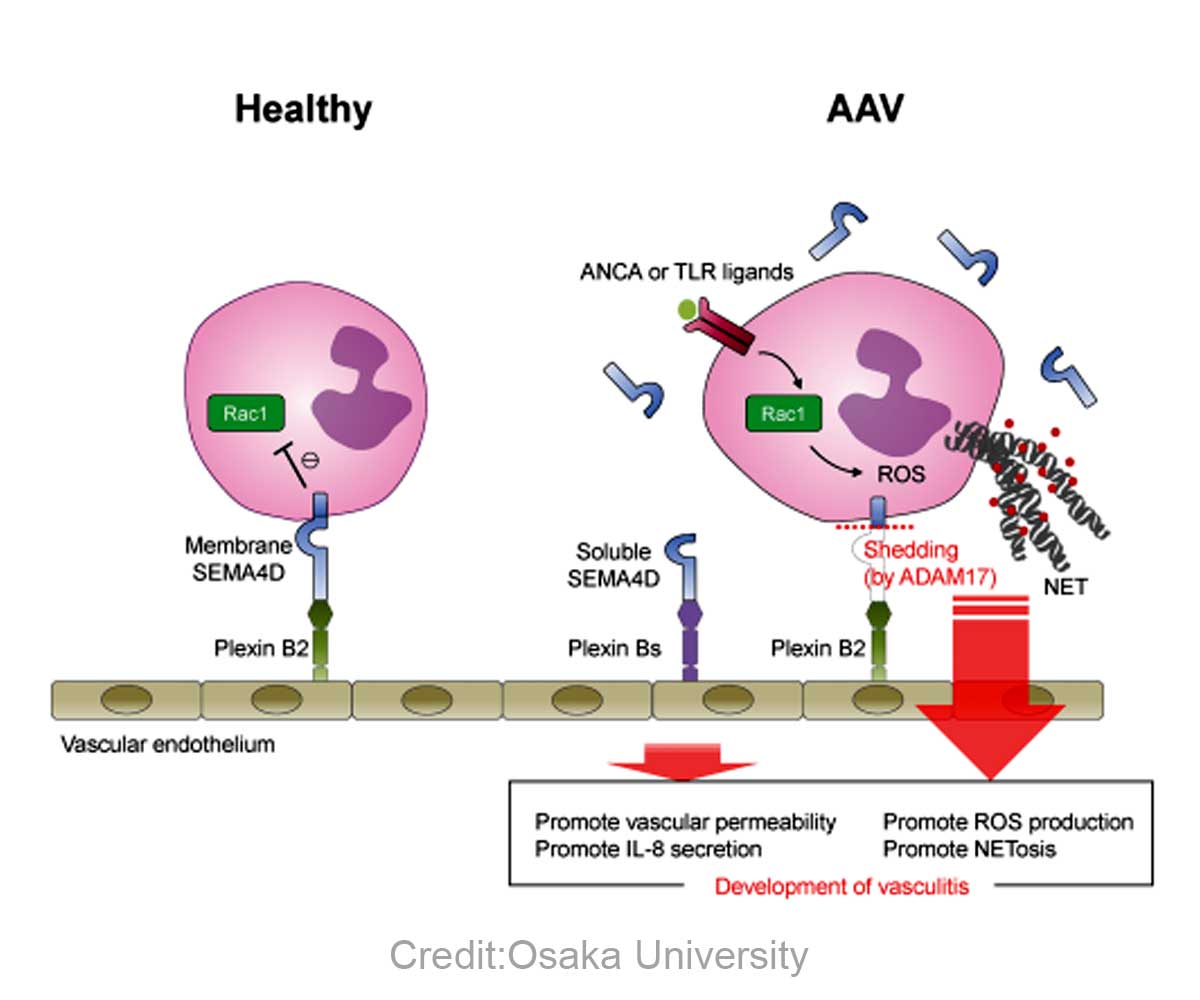
Researchers at Osaka University have now shown that SEMA4D normally restrains neutrophil activation, but that it is broken down in AAV patients, leading to inflammation and disease.
The team found that expression of the soluble form of SEMA4D was much higher in serum from patients with AAV than in healthy controls, and that levels increased with worsening disease severity. This suggested that SEMA4D could be used as a new clinical marker of AAV, which might improve the accuracy of diagnosis given that current marker expression does not always match disease activity.
To investigate the role of SEMA4D on neutrophils and in the development of AAV, the researchers knocked out its expression in mice then examined the phenotype. "In control mice, the activity of neutrophils in the form of NET production was restrained by contact with another cell type," study first author Masayuki Nishide says. "However, mice lacking SEMA4D expression missed out on this suppressive action, so their NET formation was not reduced."
Further experiments defined the membrane form of SEMA4D as a neutrophil receptor that binds the plexin B2 protein expressed by cells lining the walls of adjacent blood vessels to limit neutrophil activity. Both production of the damaging oxygen-containing molecule and NET formation were shown to be suppressed by membranous SEMA4D.
AAV is currently treated with steroid hormones to limit inflammation, or drugs that are toxic to cells. However, these findings suggest that SEMA4D has the potential to be used as a novel target in anti-AAV therapy.
Source-Eurekalert
 MEDINDIA
MEDINDIA



 Email
Email