The prenatal environment can have a lasting effect on cardiovascular and metabolic health into adulthood.
TOP INSIGHT
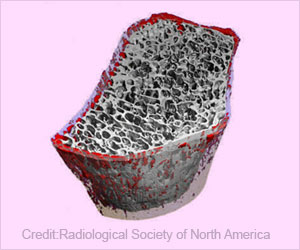
The offspring from myostatin-deficient mothers with transplanted embryos had stronger, denser bones when they grew up than mice with the same genetic makeup from osteogenesis imperfecta females.
"No cure exists; however, we know from previous research that the prenatal environment can have a lasting effect on cardiovascular and metabolic health into adulthood. We studied whether bone health of mice could be improved by optimizing the environment within the womb."
Myostatin is a protein that limits muscle growth. However, exercise causes myostatin levels to decrease - which is good because it allows muscle tissue to develop, Phillips said. Increased muscle tissue results in stronger bones. In the study, Phillips and her team decreased maternal myostatin levels to see if this would increase bone strength in offspring with osteogenesis imperfecta.
Using mice with brittle bone disease, the MU researchers were able to identify the female as responsible for offspring bone health. The team also confirmed that female mice deficient in myostatin had offspring with stronger bones.
"The third part of our study really confirmed our initial hypothesis," said Laura Schulz, associate professor of obstetrics, gynecology and women's health at the MU School of Medicine and a senior author of the study.
"Humans achieve 90% of their peak bone mass by age 19," Phillips said. "To approximate this timeframe with mice, we re-evaluated their bone strength and density four months after birth. In each case, the mice with stronger, denser adult bones were those whose fetal development involved females deficient in the protein myostatin. This finding shows that the environment within the womb affects bone development not only at birth, but into adulthood."
"The intrauterine environment is important to bone health," Schulz said. "For parents with osteogenesis imperfecta, we may be able to reduce the severity of their unborn child's disease through prenatal treatment. This also may be true for reducing the instances and severity of other bone diseases."
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email










