Researchers managed to visualize protein interactions at nanometer spatial resolution in live cells by using light switchable proteins.
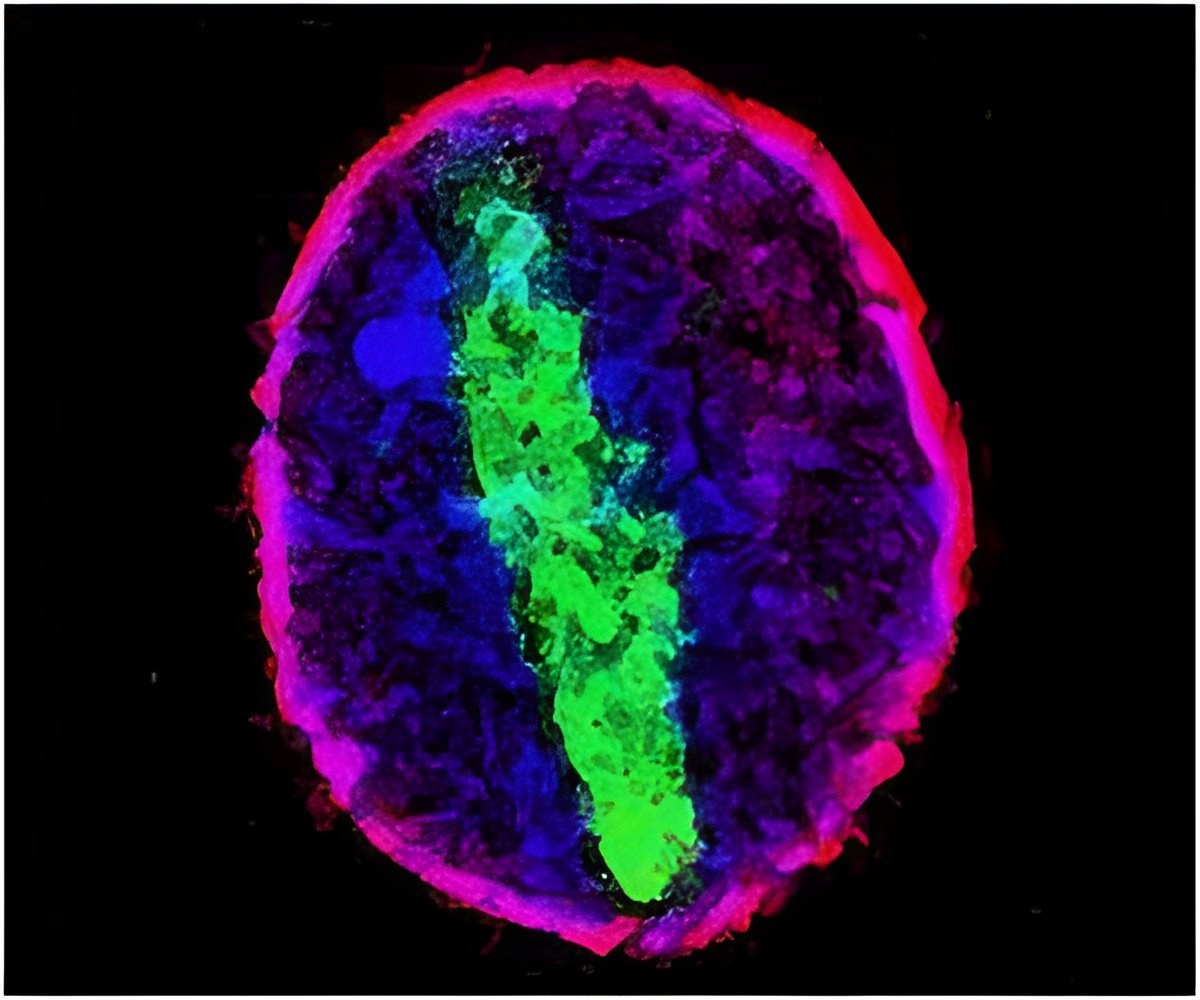
This new method uses photoactivatable complementary fluorescent proteins (PACF) to observe and quantify protein-protein interactions in live cells at the single molecule level. Yao and Xia introduced two EB proteins into cells, one with half of a photoactivatable green fluorescent protein (PAGFP), and one with the other half of PAGFP. These PAGFP pieces will only fluoresce if the EB proteins are in a complex together and photoactivated. A different wavelength of light can also be used to switch them off. The researchers could assemble super-resolution images of protein complexes by activating and then bleaching subsets of EB molecules.
The researchers opine that their technique has revealed a critical role for a previously uncharacterized EB1 linker region in tracking microtubule plus-ends in live cells. Precise localization of dynamic microtubule plus-end hub protein EB1 dimers, and their distinct distributions at the leading edge and cell body of migrating cells can be determined with the help of this technique, which can be applied to the study of other protein complexes in unprecedented detail.
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email






