A new study suggests that while kidney disease patients can successfully lose weight through weight loss surgery, majority of them do experience significant side effects.
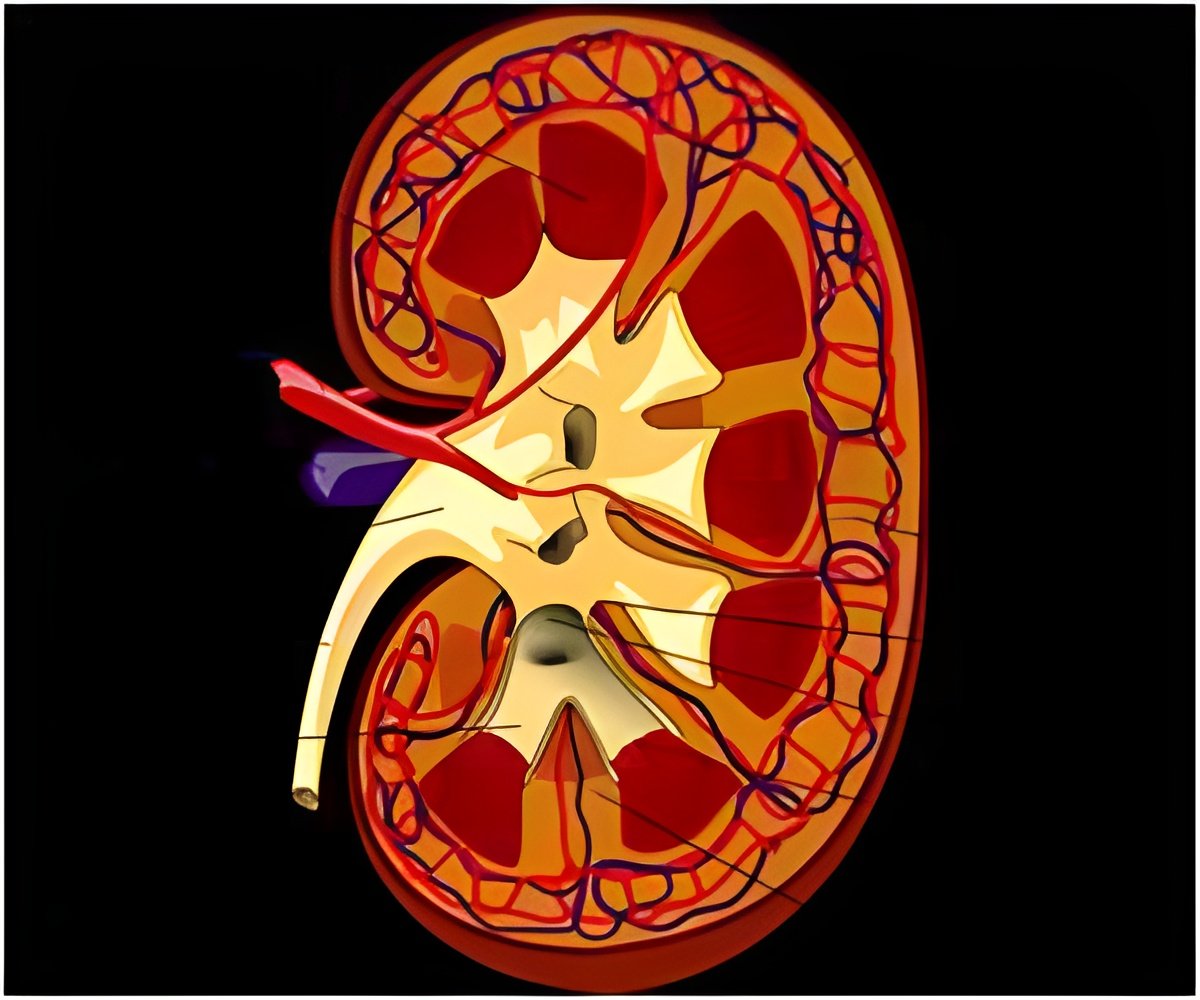
Information obtained from the 74 patients'' medical records revealed that across all forms of surgery, excess weight was lost in 61% of patients one year post-surgery. There were 16 adverse events, including two deaths (3%) related to surgical complications. Acute kidney injury was most frequent (4%), followed by leak (3%), acidosis and elevated blood potassium levels (3%), post-operative chest infection (3%), vitamin B12/iron deficiency (3%), fistula/graft failure (3%), and heart attacks (1%). An additional four deaths occurred during the study period, including two related to cancer.
"While bariatric surgery is effective for weight loss in obese patients with chronic kidney disease, the adverse event and mortality rates are high," the authors concluded. "Identification of risk factors for adverse events and investigation of non-surgical alternatives remain priorities."
Study: "Safety and Efficacy of Bariatric Surgery in Obese Patients with CKD: The London Renal Obesity Network (LonRON) Experience" (Abstract 3076)
Disclosures: Helen L. MacLaughlin is a consultant for Pentec Health. Iain C. Macdougall is a consultant for and receives honoraria and research funding from Amgen, Takeda, Vifor Pharma; consults for and receives honoraria from Johnson & Johnson; and receives research funding from AMAG. Nick Finer is a consultant for Vivus Inc., Novo Nordisk, and Arena; has an ownership interest in Counterweight plc; and receives honoraria from Novo Nordisk. Frederick W.K. Tam is a consultant for and receives research funding from Baxter Biosciences, and receives research funding from AstraZeneca and Glaxo Smith Kline. Jan Flint receives research funding from Shire Pharmaceuticals, British Dietetic Association, and honoraria from Shire Pharmaceuticals, Sanofi-Aventis, Fresenius, and Amgen. Andrew H. Frankel receives honoraria from Boehringer Ingelheim/Lilly Alliance Astra Zeneca.
ASN Kidney Week 2013, the largest nephrology meeting of its kind, will provide a forum for 14,000 professionals to discuss the latest findings in renal research and engage in educational sessions related to advances in the care of patients with kidney and related disorders. Kidney Week 2011 will take place November 5 - 10, 2013 in Atlanta, GA.
The content of this article does not reflect the views or opinions of The American Society of Nephrology (ASN). Responsibility for the information and views expressed therein lies entirely with the author(s). ASN does not offer medical advice. All content in ASN publications is for informational purposes only, and is not intended to cover all possible uses, directions, precautions, drug interactions, or adverse effects. This content should not be used during a medical emergency or for the diagnosis or treatment of any medical condition. Please consult your doctor or other qualified health care provider if you have any questions about a medical condition, or before taking any drug, changing your diet or commencing or discontinuing any course of treatment. Do not ignore or delay obtaining professional medical advice because of information accessed through ASN. Call 911 or your doctor for all medical emergencies.
Founded in 1966, and with more than 14,000 members, the American Society of Nephrology (ASN) leads the fight against kidney disease by educating health professionals, sharing new knowledge, advancing research, and advocating the highest quality care for patients.
Source-Newswise
 MEDINDIA
MEDINDIA

 Email
Email










