Italian doctor Paolo Zamboni has taken to Canada his campaign for surgery of blocked neck veins as a treatment option for multiple sclerosis (MS).
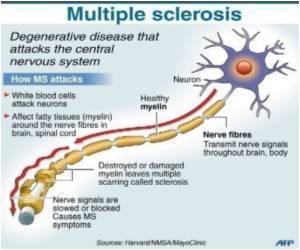
He maintains the procedure, also known as liberation therapy, has resulted in better cognitive and motor function. The endovascular surgical procedure involves inserting a tiny balloon or stent into blocked veins in order to improve the flow of blood out of the brain and spinal cord.
His quest for an effective treatment of MS started when his wife Elena Ravalli became a victim back in 1995. Though seemingly healthy at 37 years, she began to experience strange attacks of vertigo, numbness, temporary vision loss and crushing fatigue. They were classic signs of multiple sclerosis, a potentially debilitating neurological disease.
Dr. Paolo Zamboni, a professor of medicine at the University of Ferrara in Italy, set out to solve the mystery of MS – an illness that strikes people in the prime of their lives but whose causes are unknown and whose effective treatments are few.
Using ultrasound to examine the vessels leading in and out of the brain, Dr. Zamboni made a startling find: In more than 90 per cent of people with multiple sclerosis, including his spouse, the veins draining blood from the brain were malformed or blocked. In people without MS, they were not.
His research made him conclude MS was not an autoimmune condition, but a vascular disease. Zamboni calls the condition "chronic cerebrospinal venous insufficiency," or CCSVI.
More striking still was that, when Dr. Zamboni performed a simple operation to unclog veins and get blood flowing normally again, many of the symptoms of MS disappeared. The procedure is similar to angioplasty, in which a catheter is threaded into the groin and up into the arteries, where a balloon is inflated to clear the blockages. His wife, who had the surgery three years ago, has not had an attack since.
On June 11, 2010, the US National MS Society and MS Society of Canada announced a commitment of over $2.4 million to support 7 initial CCSVI grants to determine the role of CCSVI in the MS disease process. Further studies are also taking place at centers including University at Buffalo Medical Center and the Center for Vascular Awareness in Albany, NY.
The National MS Society pf the US said it shares the public’s sense of urgency to expeditiously advance any lead that has the potential of stopping, repairing or preventing MS. Accordingly, the Society is pursuing this new and potentially promising research direction by committing to fund expanded research on CCSVI in MS.
The MS Society of Canada announced last week that it is spending $700,000 on four studies to determine if there is a link between blocked veins and MS, before it will support surgical treatment. The society says researchers elsewhere in the world haven't been able to reproduce Zamboni's findings.
The society wants more people assessed for blockages that are objectively evaluated by doctors, said spokeswoman Karen Lee.
Research needs to include people who receive vein treatment to see whether they improve, just as a drug therapy would be compared with a placebo, said Dr. Robert Maggisano, a vascular surgeon at Sunnybrook Health Sciences Centre in Toronto.
"We're wasting time and money, and certainly not helping our people, who are going overseas to get treatment, when we should be able to do the studies in a blinded fashion within Canada," Maggisano said.
Alain Beaudet, president of the Canadian Institutes of Health Research, said the institute is seeking proposals for a blinded, randomized control trial that includes the procedure. The deadline for proposals is mid-August.
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email







