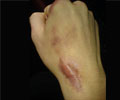
Integra LifeSciences has revealed that its Integra Flowable Wound Matrix device has received European CE Mark Certification.
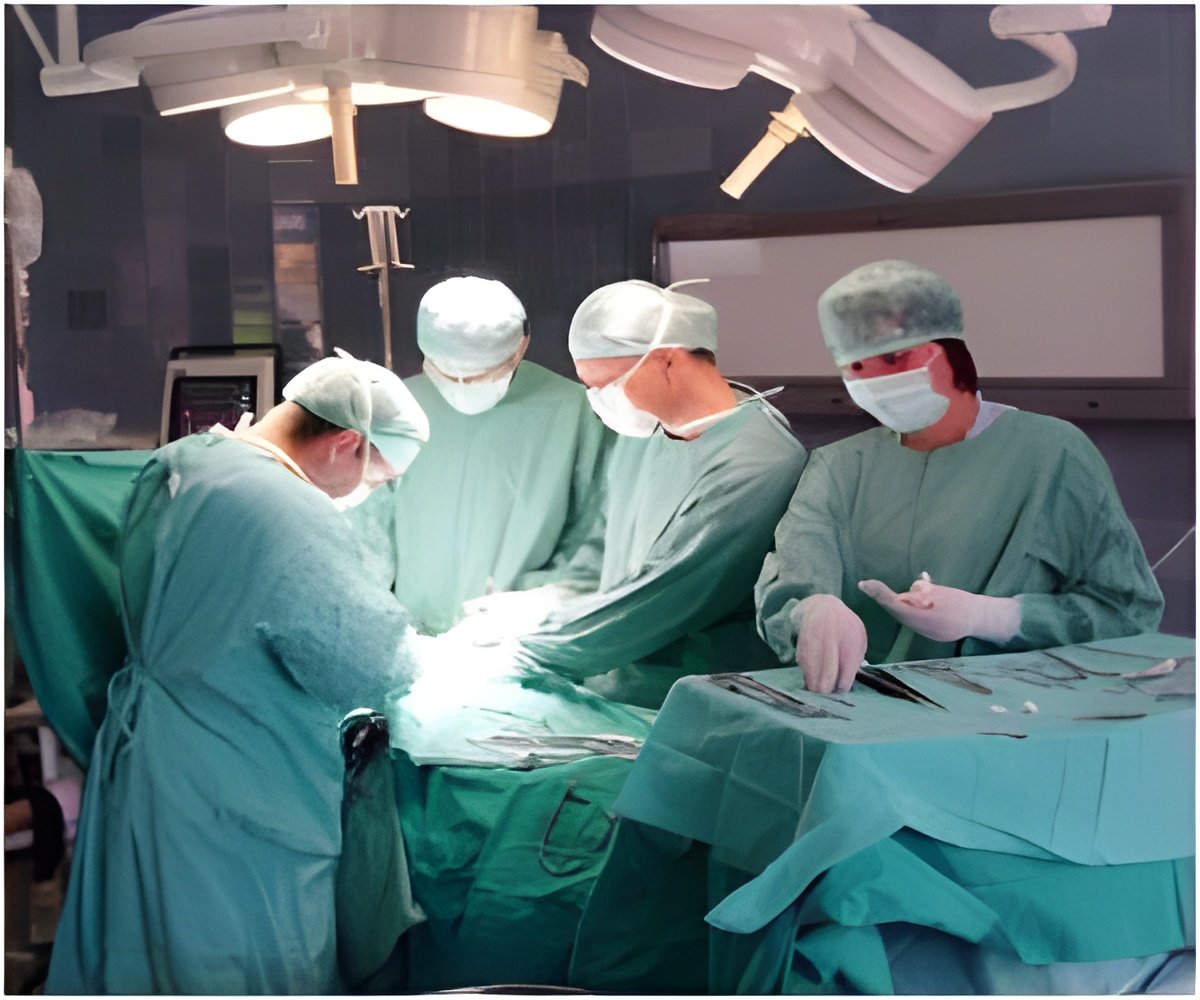
Stating that the device offers new options for both the doctors and patients, Integra’s Debbie Leonetti said, “Tens of thousands of patients in Europe suffer from chronic foot and leg ulcers that progress to deep or tunneling wounds, and they face serious risk of amputation. Integra's Flowable Wound Matrix now offers surgeons, and patients, a new option for managing these hard-to-access wounds.”
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email