CINCINNATI – Inhibiting an enzyme in the brains of newborns suffering from oxygen and blood flow deprivation stops a type of brain damage that is a leading cause of cerebral palsy,
Researchers at Cincinnati Children's Hospital Medical Center have opined that inhibiting an enzyme in the brains of newborns suffering from oxygen and blood flow deprivation stops a type of brain damage that is a leading cause of cerebral palsy, mental retardation and death.
Reporting their results in the Journal of Neuroscience, the scientists show blocking the brain enzyme, tissue-type plasminogen activator (tPA), prevents progressive brain damage triggered by the lack of oxygen and blood supply. The experimental pre-clinical treatment involved putting a naturally occurring substance called plasminogen activator inhibitor-1 (PAI-1) into the brains of newborn rats, said Chia-Yi Kuan, M.D. PhD, senior investigator on the study and a researcher in the divisions of Developmental Biology and Neurology at Cincinnati Children's.Besides demonstrating the brain's plasminogen activator system plays a pivotal role in neonatal cerebral hypoxic-ischemic brain injury, Dr. Kuan said the study also shows this system may be a promising therapeutic target in infants suffering hypoxic-ischemic encephalopathy (HIE). Identification of a treatment target is a vital step to finding better ways to treat newborns with HIE.
"Not only is hypoxic-ischemic encephalopathy an important cause of perinatal mortality and permanent neurological morbidities, but there are no specific medications against HIE in current medical practice," explained Ton J. DeGrauw, M.D., Ph.D., director of Neurology at Cincinnati Children's. "This is why the findings of this study may have important clinical implications because, in a rodent model of HIE, they that show inhibiting plasminogen activators in functional areas of the brain is powerful strategy for brain protection."
Earlier studies have pointed to the role certain brain proteases, or enzymes, play in adult brain injury following stroke, but very little has been known about what these enzymes do in neonatal cerebral hypoxic-ischemia, according to the researchers. The enzyme in this case, tPA, normally breaks down proteins and other molecules to eliminate blood clots.
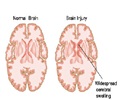
The current study, posted on the journal's website this month, included one-week-old rats in which brain hypoxia-ischemia was induced. The researchers found that hypoxia-ischemia leads to increased tPA activity. The enzyme then damages the brain blood vessels and the blood-brain barrier. The blood-brain barrier is a protective system designed to prevent invasions of blood-borne materials, in particular inflammatory cells or potential contaminants, into the central nervous system.
Dr. Kuan and his colleagues report that elevated tPA activity is triggered earlier in brain hypoxia than other proteases, particularly matrix metalloproteinases, in the progressive molecular process that leads to brain injury. This makes it a better target for therapeutic intervention to shut down the injury process early.
The researchers found that injection of PAI-1 greatly reduced the activity of both tPA and another brain enzyme, urokinase-type plasminogen activator. It also blocked hypoxic-ischemia-induced activation of matrix metalloproteinases and damage to the blood-brain barrier. The researchers also applied magnetic resonance imaging tests and microscopic analysis of brain tissues, which showed that PAI-1 treatment greatly reduced brain edema, axonal degeneration and the loss of brain tissue.
In human newborns, hypoxic brain injury usually occurs right before, during or shortly after birth, affecting two to four of every 1,000 births. The causes are mostly unknown, although some studies point to the possibility of problems with the placenta, maternal blood pressure or complications related to the umbilical cord. Close to 20 percent of newborn children with hypoxic-ischemia die and 25 percent of those who survive suffer from lifelong neurological problems.
Source-Eurekalert
TAN
 MEDINDIA
MEDINDIA


 Email
Email