Study suggests the need to improve and expand access to hydroxyurea in the U.S and internationally.
TOP INSIGHT
Maximizing the dose of hydroxyurea increased levels of fetal hemoglobin and reduced the odds of hospitalizations for young sickle cell anemia patients.
"Our analysis showed that using this approach, hospitalizations for the average patient fell to less than one every couple of years rather than four to six annually," said lead author Jeremie Estepp, M.D., an assistant member of the St. Jude Department of Hematology. "This frees children from the fevers, pain and other symptoms of this disease and gives them and their families more chances to enjoy childhood and adolescence."
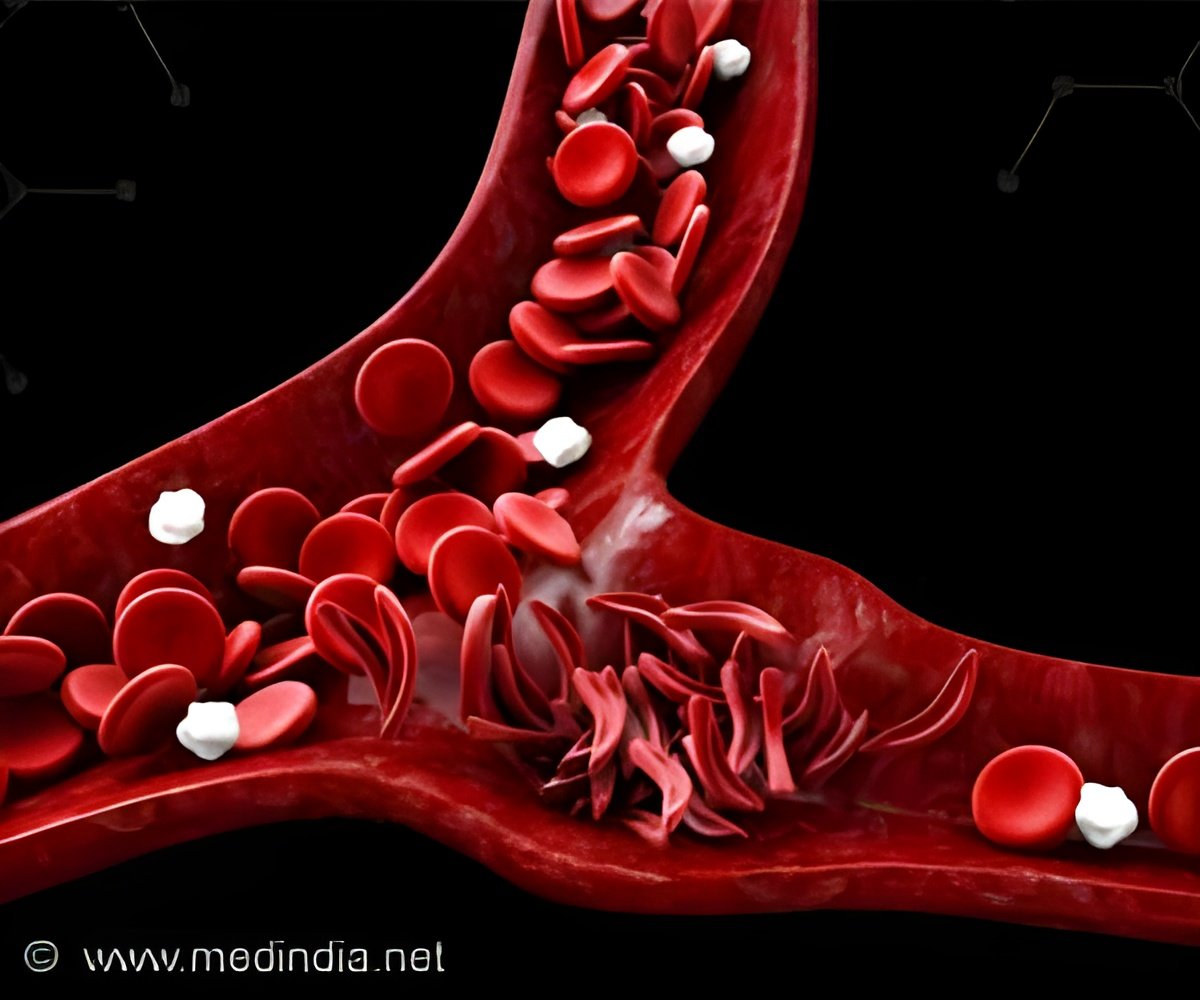
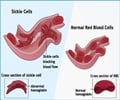
Sickle cell anemia is the most common inherited blood disorder. The disease affects about 100,000 U.S. residents and occurs most often in African-American and Hispanic individuals. They inherit a mutation that affects the hemoglobin protein that red blood cells use to ferry oxygen throughout the body. Rather than soft, pliable red blood cells that travel easily through blood vessels, the sickle cell mutation leaves patients with red blood cells that can become brittle and banana-shaped. The sickled cells clog blood vessels and lead to pain, organ damage, strokes and other symptoms and also leave patients at risk for premature death in middle age.
St. Jude pioneered the use of hydroxyurea to treat sickle cells in children. The drug is now offered to all sickle cell patients who are 9 months or older. The drug increases production of fetal hemoglobin, which functions normally in individuals with sickle cell anemia. Previously published data from HUSTLE and other studies reported that hydroxyurea was safe, reduced stroke risk and improved spleen and kidney function.
Despite hydroxyurea's proven benefits, the optimal dosing strategy for the drug has remained controversial. Also lacking has been evidence of overall improved patient outcomes if hydroxyurea is used to increase blood levels of fetal hemoglobin to more than 20 percent.
More than half the patients received a hydroxyurea starting dose of 20 milligrams per kilogram. The average dose was 27 milligrams per kilogram daily when the dose escalation process ended and ranged among patients from 13 to 35 milligrams per kilogram daily.
Average fetal hemoglobin levels in patients rose from 9.7 percent prior to treatment to 21.7 percent, a level that was sustained for the four years covered in this analysis. Overall, 60 percent of study patients attained fetal hemoglobin levels of more than 20 percent.
The odds of being hospitalized for pain or any reason were about doubled for sickle cell patients with fetal hemoglobin levels of 20 percent or less. The lower level was also associated with 2.6-times greater odds of hospitalization for the respiratory illness acute chest syndrome and about four-time greater odds of being hospitalized for fever.
"These results support a hydroxyurea dosing strategy designed to produce fetal hemoglobin levels that exceed 20 percent in an effort to decrease hospitalization of children with sickle cell disease," Estepp said. St. Jude researchers are now leading a multi-center trial to determine if toddlers with sickle cell disease would benefit from a similar dosing strategy or would respond better to a standard dose.
Estepp said the findings also underscore the need to improve and expand access to hydroxyurea in the U.S. and internationally.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email




