Central retinal vein occlusion could be treated using low cost bevacizumab drug which is effective as aflibercept drug.
Highlights
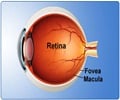
- Central retinal vein occlusion (CRVO) occurs when the vein carrying the blood flow to the retina is blocked.
- Scientists have found a cancer biologic bevacizumab drug to be as effective as aflibercept drug to treat CRVO.
- Bevacizumab drug is found to be available at a low cost with similar effects to that of aflibercept drug.
TOP INSIGHT
Bevacizumab drug found to be as effective, but less expensive than Aflibercept drug to treat central retinal vein occlusion.
Monthly eye injections after six months improved visual acuity on average from 20/100 to 20/40.
Ingrid U.Scott, M.D., M.P.H., professor of ophthalmology and public health sciences at Penn State University, said, "This head-to-head comparison of two widely used drugs for treatment of macular edema due to central retinal vein occlusion shows that both are effective in improving vision."
"The bottom line is that in patients with CRVO-associated macular edema, Avastin was as effective as Eylea in terms of visual acuity after six months of treatment."
Anti-VEGF Drugs
Anti-VEGF drugs act by making the injured blood vessels less leaky and decreases macular edema.
Bevacizumab drug is an anticancer biologic approved by the FDA. It is less expensive than aflibercept and is used off-label to treat conditions of the eye including CRVO.
Efficacy and Safety of the Anti-VEGF Drugs
The research study was conducted on 362 patients who had CRVO-associated macular edema from 66 clinical sites in the United States.
The patients were randomly assigned either Bevacizumab or Aflibercept drug for every four weeks in the six months. After six months, the research team assessed the visual, retinal thickness and side effects.
Study Findings
The study findings revealed that the average visual acuity in both the groups improved by about four lines on eye chart from 20/100 visual acuity to 20/40 visual acuity.
Frederick Ferris, M.D., director of the Division of Epidemiology and Clinical Applications at NEI, said, "This is a remarkable improvement in vision."
"On average, it more than doubles the ability to resolve fine detail, and for some patients it restores their ability to drive."
The retinal thickness was imaged using optical coherence tomography (OCT). They also found macular edema to be significant in both the groups.
There was a complete resolution of macular edema in the Aflibercept drug (54%) when compared to Bevacizumab drug (29%).
The lower proportion of the eyes which had the resolution in the bevacizumab drug did not show any poorer visual acuity outcomes for 6 months.
Further research is however continued to find the cummulative effects of the drugs and to evaluate the effects of residual edema.
Scott said, "There is little information about the long-term treatment needs of patients with CRVO-associated macular edema."
"The clinical trials that were conducted for the approval of Eylea for CRVO-associated macular edema were relatively short-term, and there have been reports of patients with CRVO requiring treatment several years out. There is important additional information that can be gained with longer follow-up of SCORE2 participants."
After six months, the side effects such as elevated intraocular pressure were low and similar in both the groups.
Sabgeeta Bhargava, Ph.D., program director at NEI, said, "SCORE2 results provide invaluable data to guide clinicians and their patients toward informed decisions about CRVO treatment."
Central Retinal Vein Occlusion (CRVO)
This type of occlusion occurs when the central vein carries the blood away from the retina is blocked. When the blood flow is disrupted, there may be a failure in the adequate levels of oxygen that reaches the retina, this could trigger the release of vascular endothelial growth factor (VEGF).
Vascular endothelial growth factor (VEGF) is found to increase the permeability of blood vessels and causes swelling in the retina resulting in a condition known as macular edema. This could be a leading cause of vision loss from CRVO.
References
- Avastin as effective as Eylea for treating central retinal vein occlusion - ( https://www.nih.gov/news-events/news-releases/avastin-effective-eylea-treating-central-retinal-vein-occlusion )
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email