A small molecule BMP inhibitor may have therapeutic potential to treat and even prevent rare bone cancer, multiple hereditary exostoses (MHE).
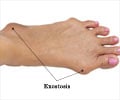
- Multiple hereditary exostoses (MHE) is a rare genetic disease which results in development of multiple bone tumors.
- The only treatment available today is surgical removal of the tumors.
- A new study identifies the mechanism of osteochondromagenesis and a potential therapy for MHE using BMP inhibitors.
One in every 50,000 people is affected with multiple hereditary exostoses (MHE). MHE is a rare genetic disease characterized by multiple bone tumors, affecting children during their growing years. Very limited treatment options and the age group that is affected emphasize the need to develop effective therapeutic strategies in treating the disease. A study conducted at the Sanford Burnham Prebys Medical Discovery Institute, USA, was successful in discovering the underlying mechanism of disease manifestation and also provide a potential therapy to treat MHE. The study was published in The Journal of Clinical Investigation. Multiple Hereditary Exostoses:
MHE is a genetic condition where people develop multiple benign (noncancerous) bone tumors called osteochondromas. MHE is also called Bessel-Hagan Disease, Multiple Osteochondroma, Familial exostoses, among others. The number and location of the tumors vary greatly from person to person. They generally form around areas of active bone growth like the knee, shoulder blades, hips and elbows, thereby restricting growth and movement. Osteochondromas are generally benign, although they may become cancerous and spread to other parts of the body. Estimates suggest that MHE has a 1 in 20 to 1 in 200 lifetime risk of becoming cancerous osteochondromas called sarcomas, which are deadly.
TOP INSIGHT
BMP inhibitor called LDN-193189 was successful in suppressing bone tumors in MHE mouse models.
Since it is a genetic condition, the disease may be passed on to children from affected parents. MHE is caused due to mutations (changes in DNA) in either EXT1 or EXT2 genes. The symptom severity is higher when the mutation is in the EXT1 gene. MHE is inherited in an autosomal dominant manner, which means that if any one parent is affected, the child has a 50% risk of inheriting the condition. The condition affects both males and females equally.
Although, affected children are born with the mutation, osteochondromas are not present at birth, however 96% of affected children develop tumors by the age of 12. Genetic testing and prenatal testing is extremely important in pregnancies with a family history of MHE.
Although there is a genetic basis for the disease, 15% of people affected with osteochondromas, do not have mutations in EXT1 or EXT2 genes. The reason for this is still unknown. This may indicate that other genetic alterations or epigenetic (genetic and environmental) factors may lead to the disease phenotype.
New Therapeutic Strategy:
Until this study, it was unclear how normal bone growth becomes abnormal in MHE patients. Professor Yu Yamaguchi at SBP said that this was essential to devise strategies to prevent, stop and maybe even reverse tumor growth.
- Stem cells of developing bone called the mesenchymal stem cell–like progenitor cells residing in the perichondrium is the cell type that gives rise to tumors in MHE.
- Enhanced or overactive Bone Morphogenic Protein (BMP) signaling in these cells is the primary cause for osteochondromagenesis (formation of osteochondromas).
- A BMP inhibitor called LDN-193189 was used to treat the MHE mouse models and it was successful in suppressing the formation of bone tumors.
References:
- Hereditary Multiple Osteochondromas - (https://ghr.nlm.nih.gov/condition/hereditary-multiple-osteochondromas#synonyms)
- Inubushi, T., Nozawa, S., Matsumoto, K., Irie, F., & Yamaguchi, Y. (2017). Aberrant perichondrial BMP signaling mediates multiple osteochondromagenesis in mice. JCI Insight, 2(15). doi:10.1172/jci.insight.90049
 MEDINDIA
MEDINDIA

 Email
Email






