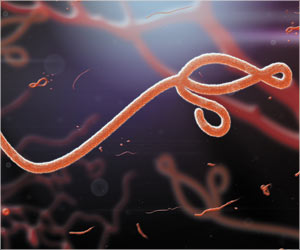
The US Food and Drug Administration has granted a fast track status to ZMapp, and this could speed its arrival on the market, reported the drug-maker.
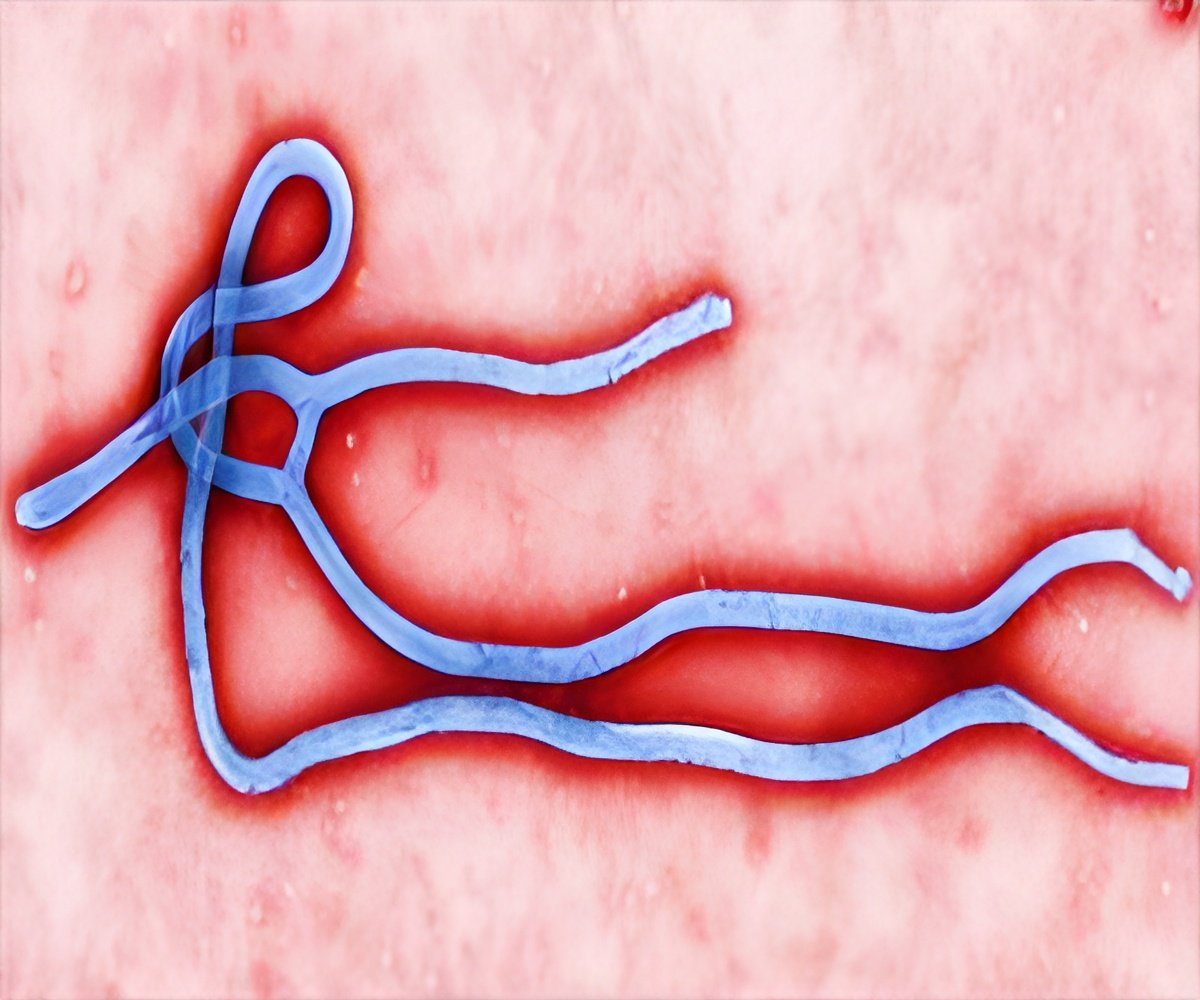
Larry Zeitlin, president of LeafBio and Mapp Biopharmaceutical, said, "The decision by the FDA is an important milestone."
Prior to getting the fast-track status, ZMapp had been granted 'orphan drug' designation, which provides financial and other regulatory incentives meant to encourage development.
Kevin Whaley, chief executive officer of LeafBio and Mapp Biopharmaceutical, based in San Diego, California, said, "We are hopeful that this step will accelerate access to ZMapp once safety and efficacy are demonstrated to FDA's satisfaction in ongoing clinical trials."
Source-AFP
 MEDINDIA
MEDINDIA
 Email
Email







