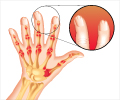
Molecular profiling techniques such as mass cytometry and single cell RNA-sequencing could predict treatment response in patients with RA.
TOP INSIGHT
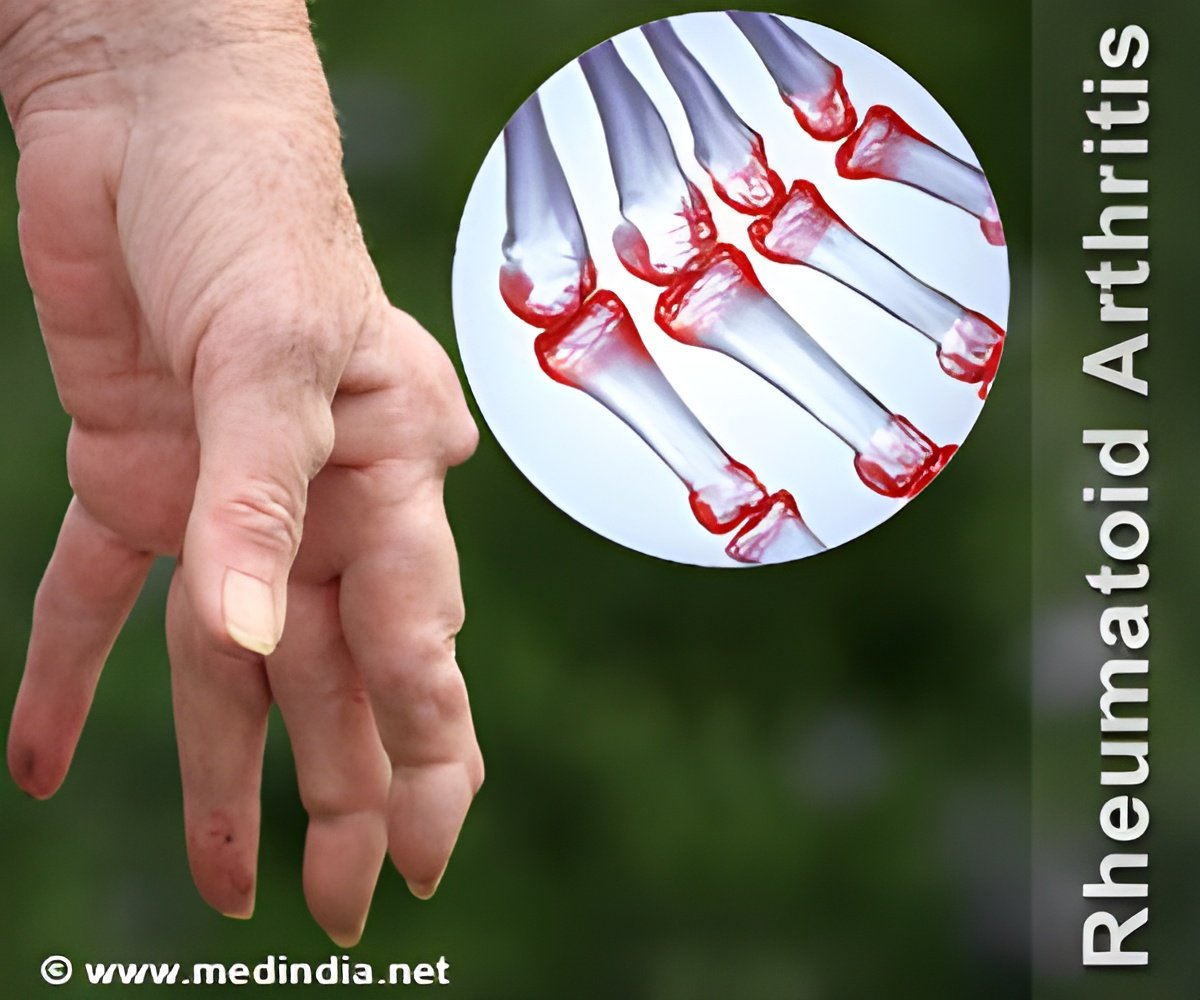
The tissues obtained from biopsies revealed significantly higher inflammation than OA tissues and a higher abundance of lymphocytes by flow cytometry.
Its RA Network of researchers has completed the first phase of a project to learn more about the disease through cellular and molecular profiling of synovial tissue and matched peripheral blood from RA patients nationwide.
"We have come a long way in our ability to treat RA patients by targeting specific pathways or cell types that contribute to joint inflammation. However, we need to better understand why certain patients do not respond to available treatments. Having biomarkers that could predict treatment response in patients with RA would be a major step forward. But, how do we identify new targets or new biomarkers given the complexity of RA?" asked Kevin Wei, Instructor at Brigham and Women's Hospital in Boston and a lead author of the study.
To answer this question, researchers used molecular profiling techniques such as mass cytometry and single cell RNA-sequencing to allow examination of 30 to 40 cell types and thousands of genes from the same patient sample.
"We believe that the ability to visualize multiple cell types and examine multiple molecular pathways in the joint target tissue will transform the way we understand RA," said Dr. Wei.
The synovial tissues were cryopreserved and shipped to a central processing center for tissue disaggregation, cell sorting, mass cytometry and RNA sequencing. Researchers then evaluated the synovial tissue quality and graded the level of synovitis.
The infiltration of lymphocytes in the tissue was significantly correlated with the histologic inflammation score, which supports the findings from the flow cytometric assessment.
Principle component analysis of the synovial flow cytometry data identified three RA arthroplasty (a common joint surgery) samples with inflammatory features that resembled those seen in the RA biopsy tissues. These arthroplasty samples also showed higher inflammation score by histology. Ongoing transcriptomic profiling of sorted synovial cells from these samples also demonstrates molecular heterogeneity in synovium from RA patients.
In the AMP-RA Network's first phase of research, the researchers found that robust cell yield from synovial tissue enables mass and flow cytometric data.
"One of the key challenges in this study was how to generate high-dimensional data from rare cell types while preserving the integrity of the tissue biology," said Dr. Wei.
"Our analyses of joint inflammation are highly consistent across multiple independent analytic methods, including histology, flow cytometry, mass cytometry and transcriptomics. Our next study will include more RA patients and longitudinal analysis of treatment response. The goal is to relate clinical course and treatment response with specific abnormal cell populations defined by mass cytometry and transcriptomics of the tissue and blood, and ultimately, to develop and test new treatment approaches."
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email