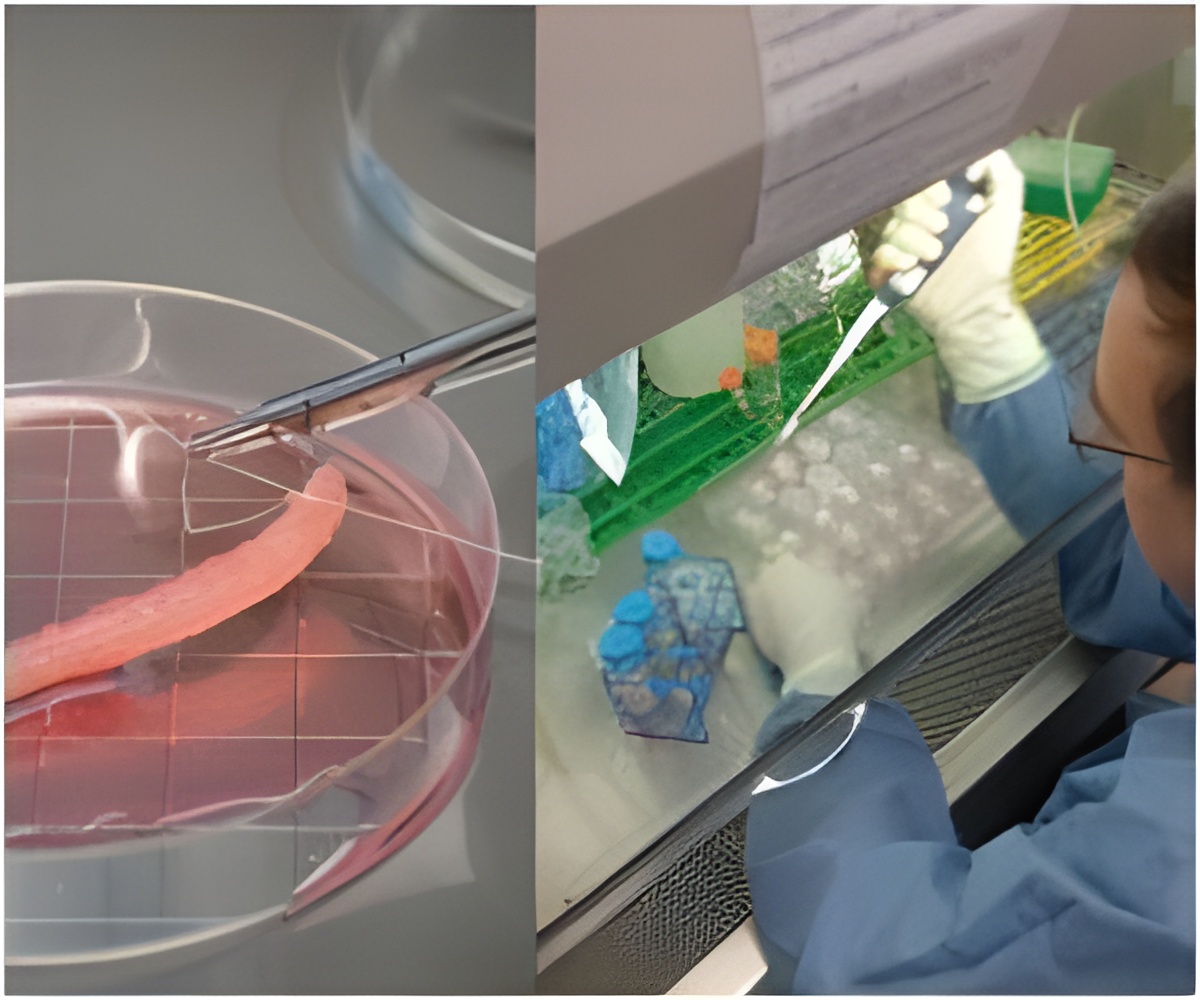
Regulation of the emerging areas of tissue engineering and regenerative medicine (TERM) is one of the key barriers to commercial and clinical success of TERM.
Countries face many of the same challenges for regulation of the emerging areas of tissue engineering and regenerative medicine (TERM). These regulatory challenges are one of the key barriers to commercial and clinical success of TERM globally. But, countries must address them in light of regional differences based on cultural, political, and economic factors, suggests a commentary published as part of a special issue of
Tissue Engineering.
David Williams, Wake Forest Institute of Regenerative Medicine, Winston-Salem, NC and Immediate Past-President,
Tissue Engineering and Regenerative Medicine International Society (TERMIS), discusses the articles contributed by authors from several Asia-Pacific countries in the context of current trends in global regulation of TERM products and services. He emphasizes the need to find the right balance between risk and benefit, and risk and innovation.
TOP INSIGHT
Countries face many of the same regulatory challenges, but they must address them in light of regional differences based on cultural, political, and economic factors.
In the article 'The Same but Different: Regulation of Tissue Engineering and Regenerative Medicine in the Context of Regional and International Standards and Expectations', Dr. Williams highlights the importance of regional differences in demographics and disease prevalence, and cultural differences related to the use of donated tissues and organs and xenogeneic materials.
"David Williams is uniquely suited to provide a balanced view of worldwide regulatory approaches to tissue engineering and regenerative medicine as a result of the significant international exposure he has had to the development and commercialization of such products," says Co-Editor-in-Chief Peter C. Johnson, Prinicipal, MedSurgPI, LLC and President and CEO, Scintellix, LLC, Raleigh, NC.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email





