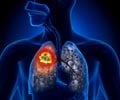
AstraZeneca has pinned hopes on durvalumab as a potential breakthrough treatment for several forms of cancer but the trials are now on hold.
TOP INSIGHT
UK-based drugmakers recently announced a $1.1bn cost-cutting plan to accelerate its shift away from mass-market primary care products and towards a new generation of specialised cancer medications.
Shares in the UK-based drugmaker have fallen more than 9.8 percent from a year earlier in the US, where it recently announced a $1.1bn cost-cutting plan to accelerate its shift away from mass-market primary care products and towards a new generation of specialised cancer medications.
The increased focus on R&D comes as it attempts to re calibrate following the recent losses of patent protection for mass-market blockbusters such as cholesterol treatment Crestor and its Nexium heartburn pill. AstraZeneca has described durvalumab as the cornerstone of its bid to develop drugs that can harness the body’s immune system to fight cancer.
Source-Medindia
 MEDINDIA
MEDINDIA

 Email
Email