Genomes of malaria parasite that affect chimpanzees and the malaria parasite responsible for the death of more than half a million children each year were found to differ.
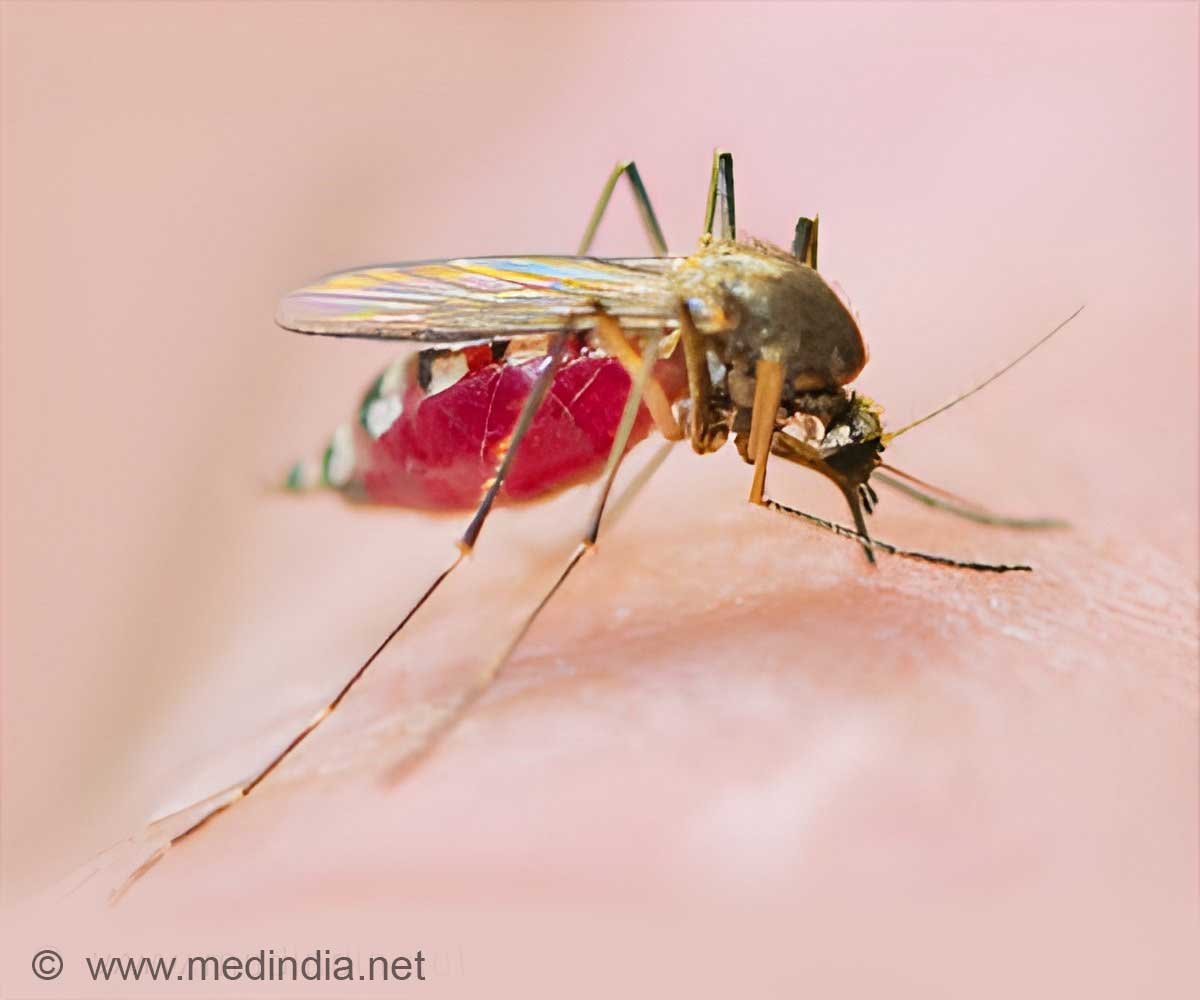
"Discovering that the key differences lie in genes responsible for red blood cell invasion reassures us that we've been looking in the right place," says Dr Thomas Otto, first author at the Wellcome Trust Sanger Institute. "Researchers have identified surface proteins as promising vaccine candidates already; and our finding adds more support, showing that it is the difference in the parasites' surface proteins that determine which host it will infect."
This is the first time that an essentially complete genome has been produced for a malaria parasite that infects such a close relative of humans. It provides the first systematic view of the differences between parasites that infect humans and those that infect our close relatives. Human malaria emerged from the Great Apes, so this comparison using chimpanzee malaria is the closest that scientists have come to a full catalogue of the changes associated with parasites switching from our primate relatives into humans.
Plasmodium parasites export proteins to the surface of red blood cells, allowing infected red blood cells to stick to the wall of blood vessels. In human malaria, the best characterised of these proteins are encoded by a highly variable family of genes, allowing the parasites to evade the host immune response and continue the infection. Surprisingly basic rules about this gene family are preserved between chimpanzee and human malaria: despite huge variation in the individual sequence of these surface antigen genes, their absolute numbers and the numbers of sub-types are remarkably preserved. By contrast, other surface antigen repertoires differed very significantly in their numbers.
"Since P. reichenowi and P. falciparum split apart, the major surface antigen gene family has not expanded or contracted; it's locked at some kind of optimised level," says Dr Matt Berriman, senior author at the Sanger Institute.
DNA used for this research was obtained by the Centre for Disease Control from a chimpanzee infected with a strain of P. reichenowi isolated in the 1950s. This chimpanzee was subsequently cured of the malaria infection. Additional blood samples were collected from orphaned infant chimpanzees infected in the wild with a similar parasite called P. gaboni. The samples were obtained from chimpanzees undergoing routine health checks at a primate sanctuary in Gabon, West Africa.
 MEDINDIA
MEDINDIA



 Email
Email







