BIOTRONIK revealed that it has received the CE approval mark for its Ilesto 7 ICD/CRT-D.
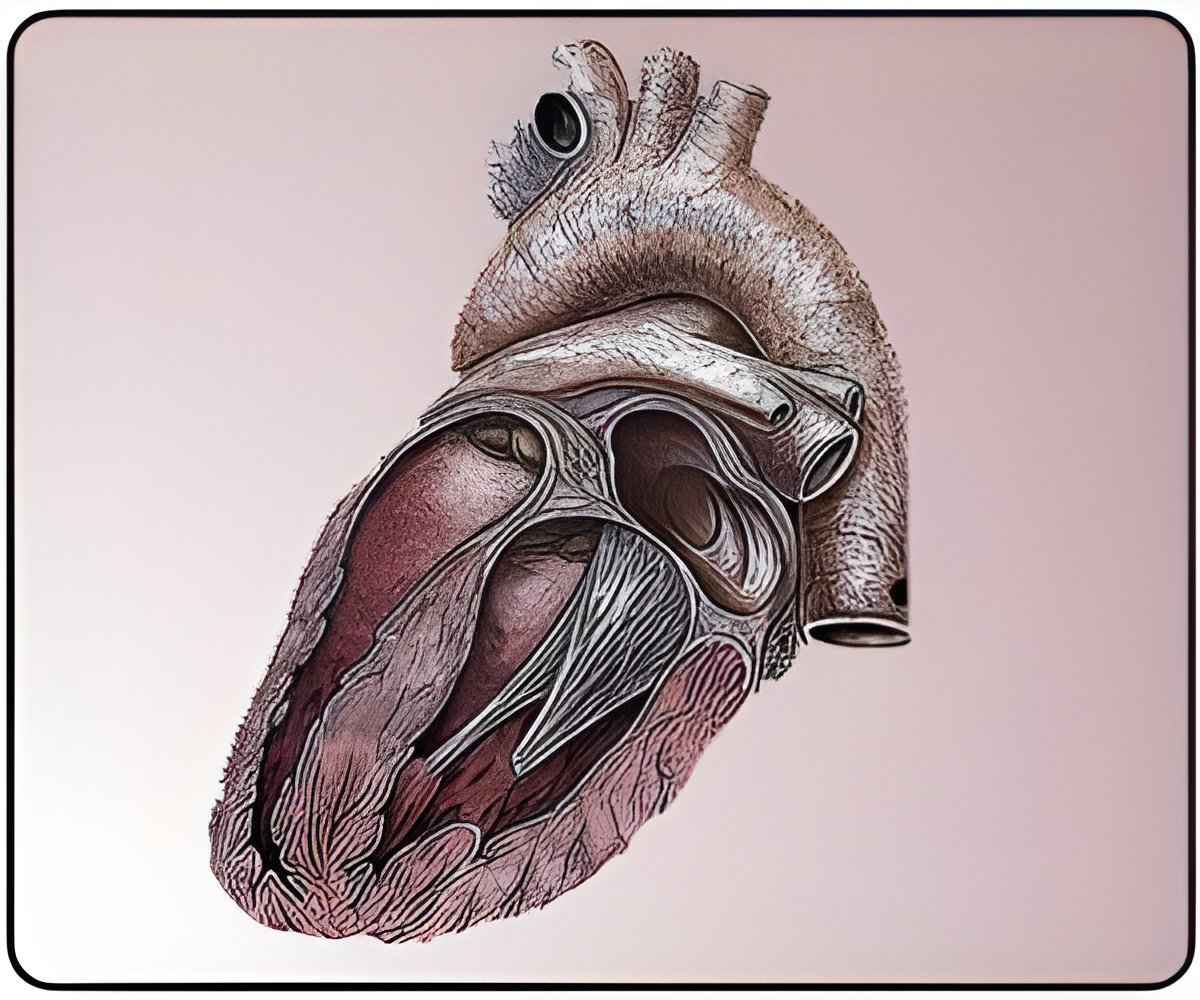
Doctors across Europe are currently performing implants of the first Ilesto 7 second-generation ProMRI devices with the patients also able to take advantage of BIOTRONIK’s in-house remote monitoring system, Home Monitoring.
Says Dr Paul Erne from Lucerne Cantonal Hospital (LUKS), Lucerne, Switzerland, who performed one of the first implants of Ilesto 7, “The Ilesto 7 series, as well as many other BIOTRONIK devices, boasts ProMRI capability and BIOTRONIK's Home Monitoring® technology. With this, we have the opportunity to offer our patients the most advanced and reliable ICD and CRT-D therapy, an enhanced quality of life, and an opportunity to live as normal a life as possible.”
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email



