The first batch of an experimental Ebola vaccine developed by Canadian scientists was sent to Switzerland for testing by the World Health Organization, reveal officials.
The vials were packed in dry ice at a temperature of minus 80 degrees Celsius (-112 degrees Fahrenheit) during shipping, officials said.
Trials of the vaccine on humans are expected to begin there at the end of the month or early in November, officials in Switzerland had said previously. Separate clinical trials of the vaccine began in the United States last week
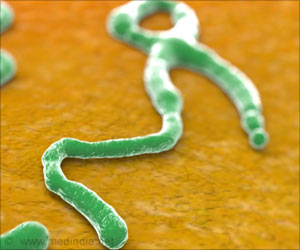
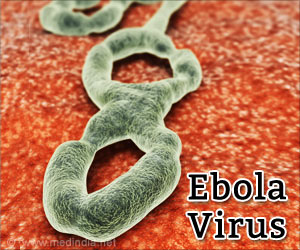
There is no licensed treatment or vaccine against Ebola, which has killed more than 4,500 people in the outbreak centered in West Africa.
However, the UN health agency has identified two experimental vaccines that have shown promising results when tested on monkeys: the Canadian VSV-EBOV, licenced by US firm NewLink Genetics, and one made by British company GlaxoSmithKline (GSK).
The WHO said last month it hoped thousands of doses of the vaccines would be ready for use in badly hit African countries by early 2015.
Advertisement
Gregory Taylor, chief public health officer, meanwhile said five rapid response teams of specialists had been placed on standby, ready to be deployed anywhere in Canada in the event of any Ebola cases in the country.
Advertisement