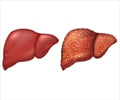
Liver is an organ that is capable of increasing by half during the day to adapt to the cycles of feeding and fasting, reveals study.

‘Liver is capable of increasing in size by almost half during the day to adapt to the cycles of feeding and fasting.’





Mammals have adapted to diurnal and nocturnal rhythms using a central clock located in the brain. The latter, which is resettled every day by the light, synchronizes the subordinate clocks present in most of our cells. In the liver, more than 350 genes involved in metabolism and detoxification are expressed in a circadian fashion, with a biological rhythm of 24 hours."Many of them are also influenced by the rhythm of food intake and physical activity, and we wanted to understand how the liver adapts to these fluctuations", says Ueli Schibler, professor emeritus at the Department of Molecular Biology of the UNIGE Faculty of Science.
The liver oscillates, but not the other organs
The mice forage and feed at night, while the day is spent resting. "In rodents following a usual circadian rhythm, we observed that the liver gradually increases during the active phase to reach a peak of more than 40% at the end of the night, and that it returns to its initial size during the day", notes Flore Sinturel, researcher of the Geneva group and first author of the study.
The cellular mechanisms of this adaptation were discovered in collaboration with scientists from the Nestlé Institute of Health Sciences (NIHS) and the University of Lausanne (UNIL) in Switzerland. Researchers have shown that the size of liver cells and their protein content oscillate in a daily manner.
The number of ribosomes, the organelles responsible for producing the proteins required for the various functions of the liver, fluctuates together with the size of the cell. "The latter adapts the production and assembly of new ribosomes to ensure a peak of protein production during the night. The components of ribosomes produced in excess are then identified, labeled, and degraded during the resting phase", explains Flore Sinturel.
Advertisement
The amplitude of the variations observed by the biologists depends on the cycles of feeding and fasting, as well as diurnal and nocturnal phases. Indeed, the fluctuations disappear when the phases of feeding no longer correspond to the biological clock, which evolved in the course of hundreds of millions of years: "the size of the liver and the hepatocytes, as well as their contents in ribosomes and proteins, remain nearly stable when mice are fed during the day.
Yet, these animals ingest similar amounts of food, irrespective of whether they are fed during the night or during the day", points out Frédéric Gachon of the NIHS, who co-directed the study.
Advertisement
If mechanisms similar to those found in mice exist in humans, which is likely to be the case, the deregulation of our biological rhythms would have a considerable influence on hepatic functions.
Source-Eurekalert