The US Food and Drug Administration has approved the marketing of Canadian pharmaceutical company Angiotech’s device.
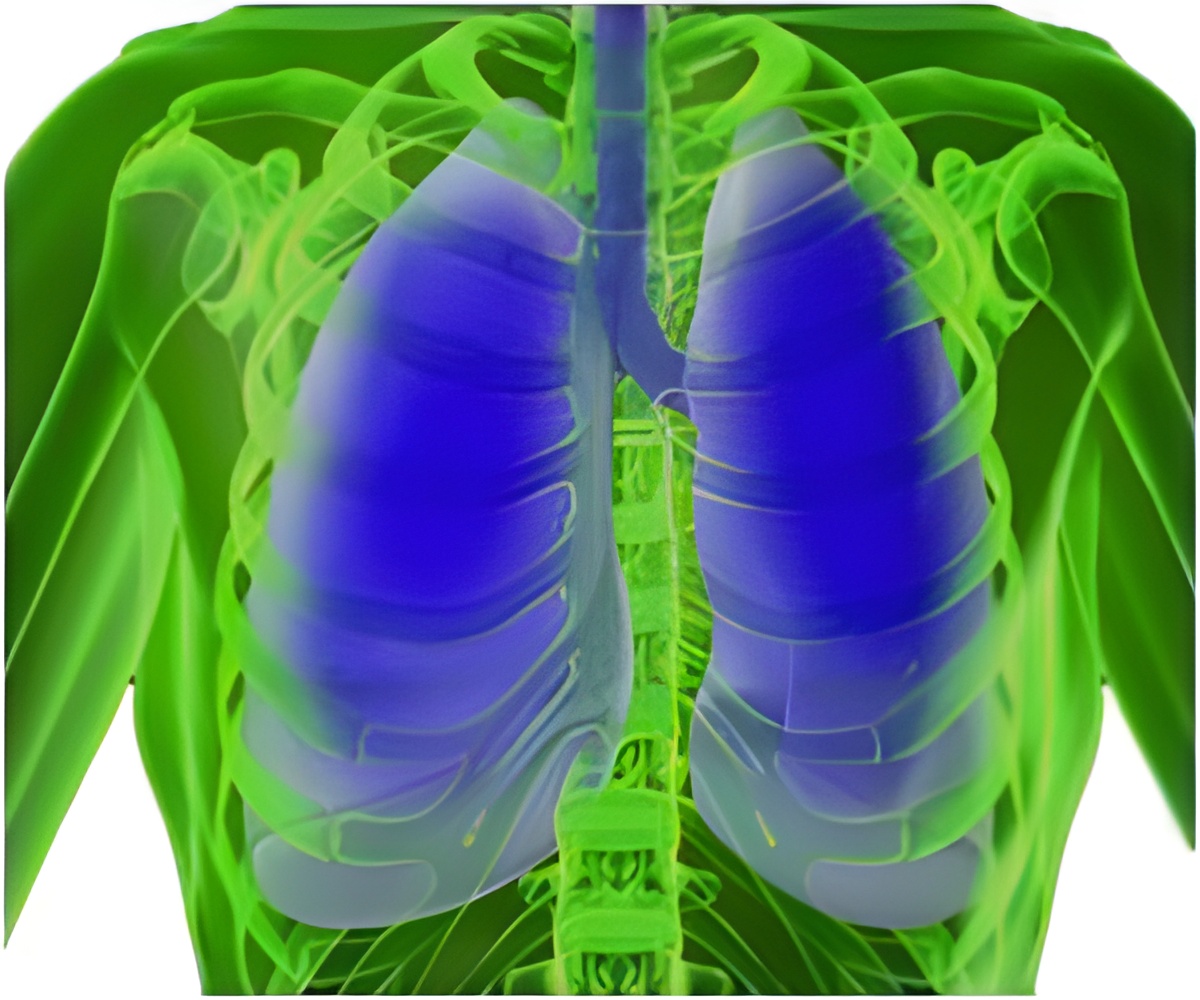
The Bio-Seal system comes with a syringe that has been filled with a hydrogel which is solidified and dried. After the biopsy is conducted, doctors attach the system to the needle and inject the hydrogel through the stylet (a slender wire) into the empty space created by removing the lung tissue for the sample.
Angiotech CEO Thomas Bailey said that the device has shown to reduce the complications caused by lung biopsies. “Bio-Seal represents a significant and proprietary addition to our broad portfolio of products for the diagnosis of cancer. This device has been shown in clinical studies to significantly reduce complication rates in patients receiving a lung biopsy procedure, which we expect will enable physicians to more aggressively pursue diagnostic options for this patient population”, he said.
Source-Medindia
 MEDINDIA
MEDINDIA
 Email
Email