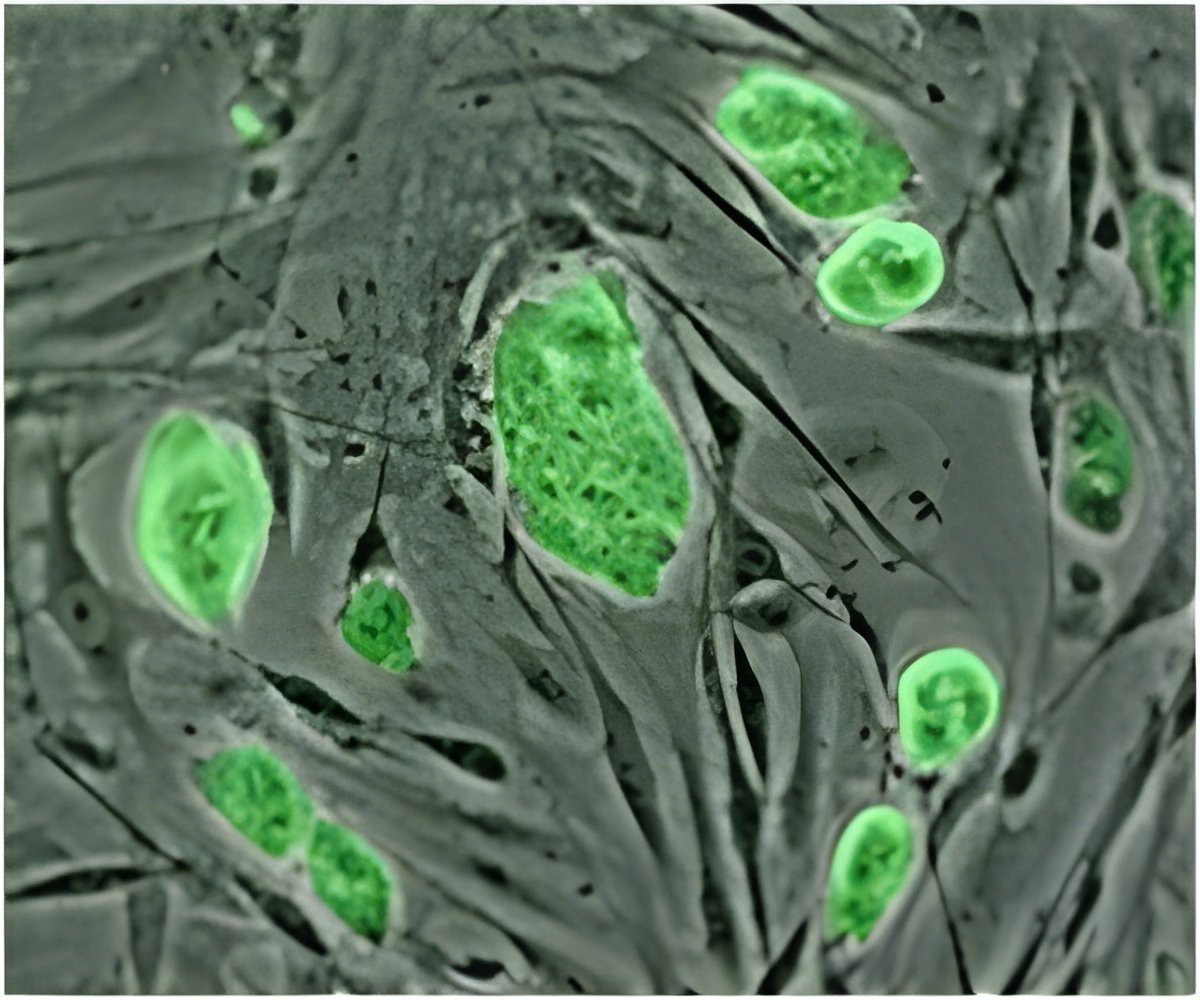
The US company's trial on patients with recent spinal cord injuries, the first on humans to be approved by the US Federal Drug Administration, began in October 2010 and four people have so far been treated.
There had been no safety issues and the decision to end the study was due to a lack of money and the complexities of getting regulatory approval.
New patients will no longer be enrolled but the progress of those already involved, who were each injected with around two million cells, will be followed.
According to the company, in the first phase of the trial, which was meant primarily to gauge safety, the therapy was "well tolerated with no serious adverse events".
The company also said that the stem cells "continue to hold great medical promise" but the decision was taken after a "strategic review" of costs, and it intends to focus on developing anti-cancer drugs instead.
Advertisement
The ending of the programme was greeted with anger by Daniel Heumann, a board member of the Christopher and Dana Reeves Foundation, which aims to cure spinal cord injury by funding new paths of research.
Advertisement
Those who support embryonic stem cell research believe that the field holds great promise for a host of conditions including Alzheimer's, Parkinston's, heart disease and stroke.
Embryonic stem cells can develop into any type of cell in the body and researchers hope one day they might be used to replace or repair damaged tissue, but the research is controversial because it requires the destruction of human embryos, and opponents say it violates the sanctity of life.
Source-ANI