- Mucopolysaccharidosis type II - (http://ghr.nlm.nih.gov/condition/mucopolysaccharidosis-type-ii)
- Mucopolysaccharidosis IV - (http://rarediseases.org/rare-diseases/morquio-syndrome/)
What are Mucopolysaccharidoses?
Mucopolysaccharidosis (MPS) are a group of genetic metabolic disorders that manifest due to the absence or malfunctioning of specific enzymes required to process molecules called glycosaminoglycans.
Glycosaminoglycans are long chains of complex sugar molecules in cells that participate in the formation of bone, cartilage, tendons, cornea, skin and connective tissue. These complex molecules are also found in the fluid lubricating joints.
People with MPS do not produce enough of 1 of the 11 enzymes needed to break down glycosaminoglycans, or they produce altered enzyme that is less effective. This leads to glycosaminoglycan buildup in the cells, blood and connective tissue and consequent cellular damage. The cellular damage manifests as symptoms like abnormal facial features and appearance, impaired physical ability, organ malfunctioning and in some cases, cognitive impairments and developmental delay.
MPS is a part of the lysosomal storage disorders family. These disorders are caused by malfunctioning of lysosomes. Lysosomes digest carbohydrates, proteins, etc. in the cell through enzymes. Lysosomal storage disorders occur when the enzymes are either totally absent or insufficient or malfunctioning, thus resulting in accumulation of certain molecules, which can cause tissue damage.
What are the Types of MPS?
MPS is classified into seven clinical types:
- MPS type I: MPS type I occurs due to lack of or insufficient production of the enzyme, alpha-L-iduronidase. Based on the severity of symptoms, it is divided into three subtypes - Hurler, Hurler-Scheie and Scheie, with Hurler being the most severe.
- In the less severe forms of MPS I, an onset of symptoms may occur by the age of 5 years. In some cases, these less severe forms are diagnosed only after the age of 10 years. On the other hand, in the severe form of Hurler syndrome, developmental delay, is seen by the end of the first year.
- Children with MPS I present a range of symptoms like short stature, short body trunk, abnormal facial features, corneal clouding, vision impairment, joint and bone issues, organ enlargement, breathing difficulties, cardiac issues and mental decline.
- MPS type II (Hunter syndrome): MPS II is caused either by a total lack or insufficient production of the enzyme, iduronate sulfatase. Individuals with MPS II have similar clinical symptoms as those with MPS I, though in lesser severity. In the less severe form of MPS II, diagnosis may be as late as in the twenties. People with mild or moderate MPS II often live beyond their 50s.
- MPS type III (Sanfillipo syndrome): There are four subtypes of MPS III based on the missing or insufficient enzyme:
- MPS IIIA is caused by missing or insufficient enzyme heparan N-sulfatase.
- MPS IIIB is caused by missing or insufficient enzyme alpha-N-acetylglucosaminidase.
- MPS IIIC is caused by missing or insufficient enzyme acetyl-CoAlpha-glucosaminide acetyltransferase.
- MPS IIID is caused by missing or insufficient enzyme N-acetylglucosamine 6-sulfatase.
- Clinical symptoms in all the four subtypes are similar although people with type A experience faster degeneration. Patients with MPS III usually present with severe neurological symptoms. Children present with hyperactivity, inability to sleep beyond a few hours, vision impairment, hearing loss, seizures and aggressive behavior. Motor and mental skills are usually delayed. Muscle degeneration may lead to an inability to walk by the age of 10 in some cases. Some children develop tonsils and adenoids which require surgical removal. Frequent respiratory infections are common.
- MPS type IV (Morquio syndrome): MPS type IV is caused by missing or insufficient enzymes, N-acetylgalactosamine 6-sulfatase (Type A) or beta-galactosidase (Type B). Both these enzymes are required to break down the keratan sulfate sugar chain. MPS IV is usually diagnosed between the ages 1-3 years. Children develop bone deformities which can compress nerves, joint pains, vision impairment, respiratory and cardiac issues. Intellectual ability is generally normal unless individuals develop hydrocephalus which is left untreated. Type IVB is milder, though both type IV-A and type IV-B share similar clinical presentation.
- MPS type VI (Maroteaux-Lamy syndrome): MPS type VI is caused by the insufficient enzyme, N-acetylgalactosamine 4-sulfatase. MPS VI is similar to MPS I with respect to clinical symptoms. However, children with MPS VI have normal intelligence. Some of the shared symptoms include clouding cornea, hearing loss, nerve compression, stunted growth, hernias and heart disease.
- MPS type VII (Sly syndrome): MPS type VII is caused by the missing or insufficient enzyme, beta-glucuronidase. This is the least common forms of MPS. In rare cases, children born with MPS VII have hydrops fetalis where excess fluid is retained in the body. This is fatal unless treated on time. In children with mild to moderate MPS VII, corneal clouding and loss of peripheral and night vision is common. Other symptoms include short stature, stunted growth, hernias and respiratory issues.
- MPS type IX: MPS type IX is caused by the missing or insufficient enzyme, hyaluronidase. As of 2001, there was only one case of type IX reported. Symptoms included painful swelling of joints. Imaging indicated bone erosion and soft-tissue masses. Facial abnormalities, short stature and stunted growth were present.
All the clinical types and subtypes of MPS are inherited in an autosomal recessive pattern with the exception of MPS II, which is X-linked recessive. In an autosomal recessive inheritance pattern, the individual inherits two copies of the defective gene, one from each parent. The gene in this case is located on one of the 22 pairs of chromosomes called autosomes. MPS type II is inherited through the X-chromosome, which is one of the sex chromosomes. Since women have two X-chromosomes, they do not manifest conditions with are X-linked recessive, but can pass on the defective gene to the son, who has only one X-chromosome and therefore manifests the condition.
What are the Symptoms of Mucopolysaccharidoses?
Symptoms may be mild, moderate or severe depending on the type of MPS. All individuals do not present with mental retardation or developmental delays. There is a common cluster of symptoms occurring across clinical types and subtypes of MPS:
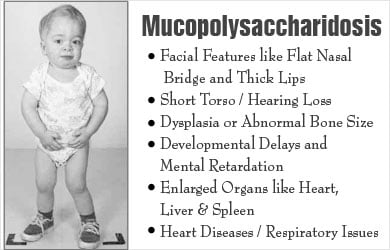
- Facial features like flat nasal bridge and thick lips
- Short torso
- Dysplasia or abnormal bone size or shape
- Hearing loss
- Corneal clouding and vision impairment
- Hydrocephalus (fluid in the brain)
- Developmental delays and mental retardation
- Enlarged organs like heart, liver and spleen
- Respiratory issues
- Heart diseases
How are Mucopolysaccharidoses diagnosed?
Diagnosis of mucopolysaccharidoses is made based on a combination of medical history, physical examination and clinical investigations.
Urine tests indicate the presence of excess mucopolysaccharides. Enzyme assays where cultured cells or blood is tested for enzyme deficiencies is also diagnostic of the specific type of mucopolysaccharidoses.

Genetic testing is available to detect carriers of the gene. Pre-natal diagnosis is also available to detect the presence of MPS in the fetus. A procedure known as chorion villi sampling (CVS) is done between the 8th and 10th week of pregnancy. This procedure involves obtaining and testing a micro sample of the placenta.
How are Mucopolysaccharidoses treated?
There is no permanent cure for MPS and treatment is symptomatic.
MPS is treated on a case-to-case basis in providing support for patients. Symptoms are tackled and the overall aim is to keep the individual as healthy and functional as possible. For example, vision impairment can be avoided by corneal surgery to remove a layer of the cloudy cornea. Respiratory infections are treated with antibiotics.
A diet with limited amounts of milk, sugar and dairy products has been helpful in some cases. Nutritional supplements may be given to maintain strength.
Enzyme replacement therapy (ERT) is being used for MPS I, MPS II and MPS VI. This is helpful in reducing pain and neurological symptoms.
Bone marrow transplant (BMT) and umbilical cord blood transplant (UCBT) have not been very successful treating MPS. These procedures also come with a range of other risks like rejection and fatal pneumonia due to suppressed immunity during the procedure.








