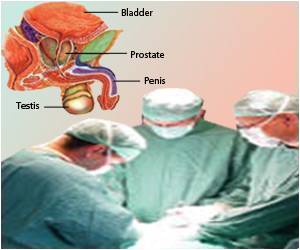
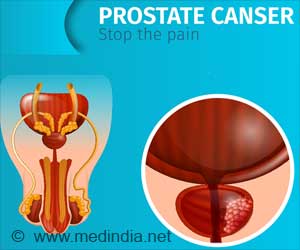
"The aggressiveness of newly diagnosed prostate cancer is variable and difficult to predict. What clinicians really need is a well-validated prognostic test that accurately predicts disease-specific mortality and can improve patient care," said Jack Cuzick, PhD., study investigator and head of the Centre for Cancer Prevention at Queen Mary University of London. "Currently, with standard clinical tools like Gleason Score or PSA, we can't adequately define prognosis. The Prolaris test provides useful new information and is highly prognostic for disease-specific death among men being conservatively treated."In this study, the goal was to validate the Prolaris test score in 761 conservatively managed prostate cancer patients diagnosed by needle biopsy.
The primary endpoint was prostate cancer death and the median follow-up time was 9.5 years. The results showed that for each one unit increase in the Prolaris score, patients had approximately double the risk of dying from prostate cancer (see table).
- Prolaris Predicts 10-Year Death Rates from Prostate Cancer
Prolaris Test Score<00-11.1-2>2
Death Rate (%)7153659
Prolaris Outperforms Upgrading Practice In the second study featured at AUA, 230 men who had pathologic Gleason Scores of either 3+4 or 4+3 were examined. The Prolaris test was performed on the samples and the rates of biochemical recurrence (BCR) were compared in each group. The results showed that there was no difference in BCR based on an assessment using Gleason Score alone. However, the Prolaris test significantly outperformed the current practice of upgrading to Gleason 3+4 or 4+3 in predicting which patients would experience biochemical recurrence after a radical prostatectomy.
These results underscore that genomic tests must be correlated with meaningful oncologic endpoints, rather than endpoints like upgrading. This finding should lead to improved initial decisions on the most effective treatment for each patient based on his personal risk of disease progression."Prolaris has been extensively validated in thousands of patients and is the most studied molecular prognostic test for prostate cancer in history," said Michael Brawer, M.D., vice president of Medical Affairs at Myriad Genetics. "Prolaris is rapidly becoming the leading test to assess the aggressiveness of prostate cancer and to help physicians select the most appropriate clinical management for patients newly diagnosed with prostate cancer."
Prolaris Is Strongest Predictor of Metastatic Prostate Cancer The third study presented at AUA evaluated the prognostic utility of the Prolaris score generated from needle biopsy in men treated with prostatectomy. Importantly, the results of this study found that the Prolaris test was the strongest predictor of metastatic disease compared to other tested clinical variables. Patients with a high Prolaris score have more than a six-fold higher risk of developing metastases compared to patients with a low score. These results confirm that Prolaris may be used at the time of disease diagnosis to better determine which patients are at highest risk and help select appropriate medical care based on their personal risk profiles.
Advertisement