Heart devices are biased toward men and do not include women, say researchers. Heart devices don't have adequate data on safety and effectiveness in women.
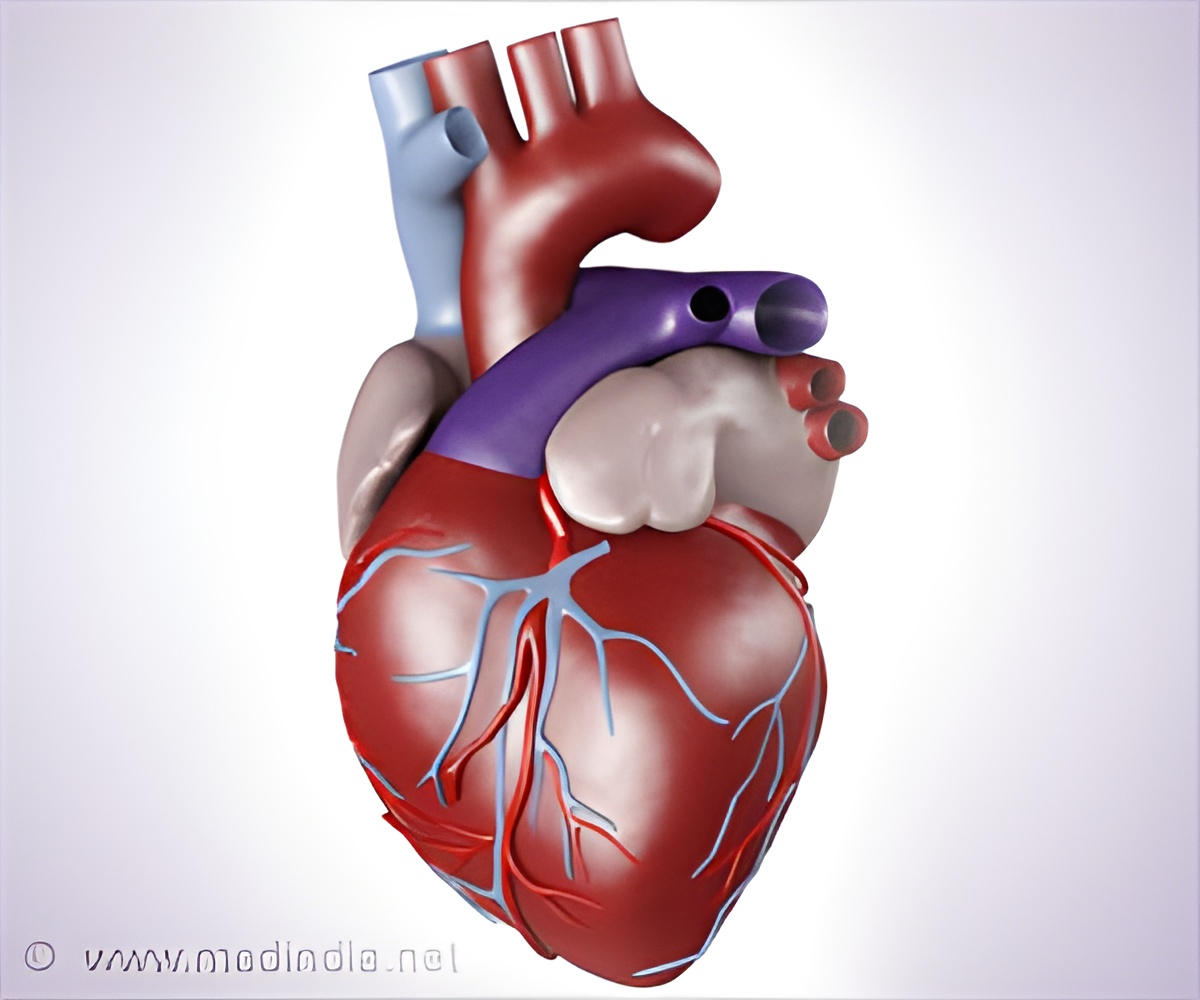
"Women in general have more procedural complications, so they tend to have more adverse events. Women tend to bleed more than men do, so we see more bleeding complications," she told AFP.
Women also tend to be an average of 10 years older than men when they get heart disease, and multiple studies have shown that risks and benefits from various devices and treatments are not the same in men and women, she said.
The US Food and Drug Administration in 1994 decided that anyone seeking approval for a new device, such as a heart valve or defibrillator, must explain in their submission whether the study subjects reflect the real-life proportion of men and women who have the condition being treated.
They must also discuss differences in how safe and effective the device was for women compared to men.
Despite this policy, Redberg found that 28 percent of studies did not report the gender of the participants at all, and among those that did, men made up an average of 67 percent of the trial.
Redberg's research examined 123 pre-market approval studies for 78 high-risk cardiovascular devices which were approved by the FDA between 2000 and 2007.
The study, co-authored by Sanket Dhruva and Lisa Bero, is published in the journal Circulation: Cardiovascular Quality and Outcomes.
It notes that Redberg is a member of the FDA Circulatory System Devices Panel, "but her comments are independent and don't necessarily reflect the views of the panel or the FDA."
The FDA said it "is aware of the under-representation of women in clinical trials, an issue which is not isolated to FDA trials," said a statement sent to AFP by spokeswoman Karen Riley.
The reasons are "complex," and may include "less willingness on the part of both females and their physicians to participate in investigational studies and invasive procedures," the FDA said.
"Certain types of heart disease are less common in women than in men, or do not appear in women until older ages, thereby limiting the number of women available for study," it added.
A new, formal guidance on how to achieve "appropriate gender representation and conducting scientifically valid analysis for sex differences" is due later this year, the FDA statement said.
Heart disease is the top killer of women in the United States, and kills more women annually than it does men.
The American Heart Association says 42 million American women and 39 million men have heart disease, and 373,000 women and 341,000 men died of it in 2006.
The AHA has also noted that women make up only 38 percent of subjects in NIH-funded cardiovascular studies.
Three in four such clinical trials do not report results according to sex, "making it difficult for researchers and clinicians to draw conclusions about their effects on women."
Redberg said the best thing for women to do is "ask directly about the data if a device is recommended to them... Ask how many women it was tested in and how the results looked in those women.
"Unfortunately, all too often we approve devices based on results in men and assume they will be the same in women. That is not a reasonable assumption, even if a device is being marketed specifically to women," she said.
Source-AFP
 MEDINDIA
MEDINDIA


 Email
Email








